CAR-T cell therapy is a T cell-based genetic engineering therapy in which T cells are collected from a patient, genetically engineered in the laboratory to express the chimeric antigen receptor (CAR), then cell expanded and given back into the patient as a "live-cell drug". In 2017, the FDA approved the first CAR T cell therapy for the market, and six CAR T cell therapies have been approved by the FDA for the treatment of B-cell leukemia, lymphoma and other blood cancers, and demonstrated a strong therapeutic effect.
The most commonly used CAR-T cell therapy, CD19-targeted CAR-T cell therapy, which targets B cells and their malignant offspring through B 9-cell highly specific and ubiquitous surface antigen CD19.
In recent years, CD19-targeted CAR T cell therapy has shown good therapeutic results for several autoimmune diseases, including systemic lupus erythematosus, systemic sclerosis, idiopathic inflammatory myopathy, and myasthenia gravie.
On March 29,2024, researchers at the Eppendorf Medical Center at the University of Hamburg, Germany, in collaboration with Kyverna Therapeutics, published a paper titled: CD19-targeted chimeric antigen receptor T cell therapy in two patients with multiple sclerosis in Med, a journal of Cell Press.
The study used the CD19-targeted CAR-T cell therapy to treat the new autoimmune disease, ——, multiple sclerosis (Multiple Sclerosis, MS).

Progressive multiple sclerosis (MS) is characterized by localized occult neuroinflammation caused by the proliferation of immune cells (including B cells) present in the central nervous system (CNS). Although inflammation can be prevented by immunomodulatory therapy in the early stage of the disease, this usually fails to prevent disease progression.
CD19-targeted CAR T cell therapies have revolutionized the treatment of hematological malignancies. Recent studies have shown the therapeutic potential of CD19-targeted CAR T cell therapies in a variety of autoimmune diseases such as systemic lupus erythematosus and myasthenia gravie.
This study reports the first treatment data of using fully human CD19-targeted CAR-T cell therapy (KYV-101) in two progressive multiple sclerosis patients. The therapy was developed by the Kyverna Therapeutics. The results showed that CAR-T cell therapy had an acceptable safety profile in both patients after administration, with the presence of CAR-T cells detected in the patient CSF, but no clinical signs of neurotoxicity were observed. Meanwhile, intrathecal antibody production in the CSF was significantly reduced after CAR-T cell infusion.
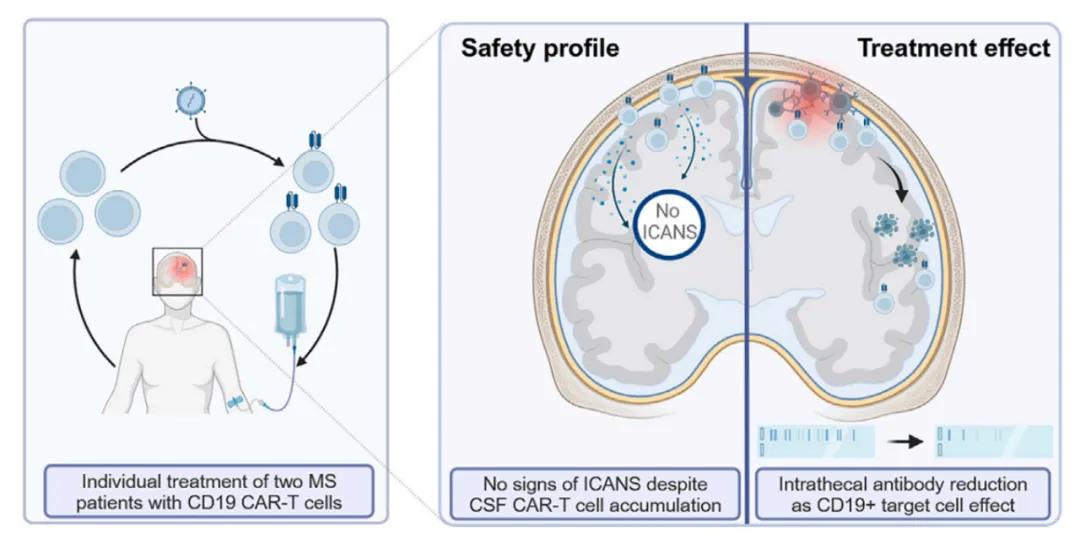
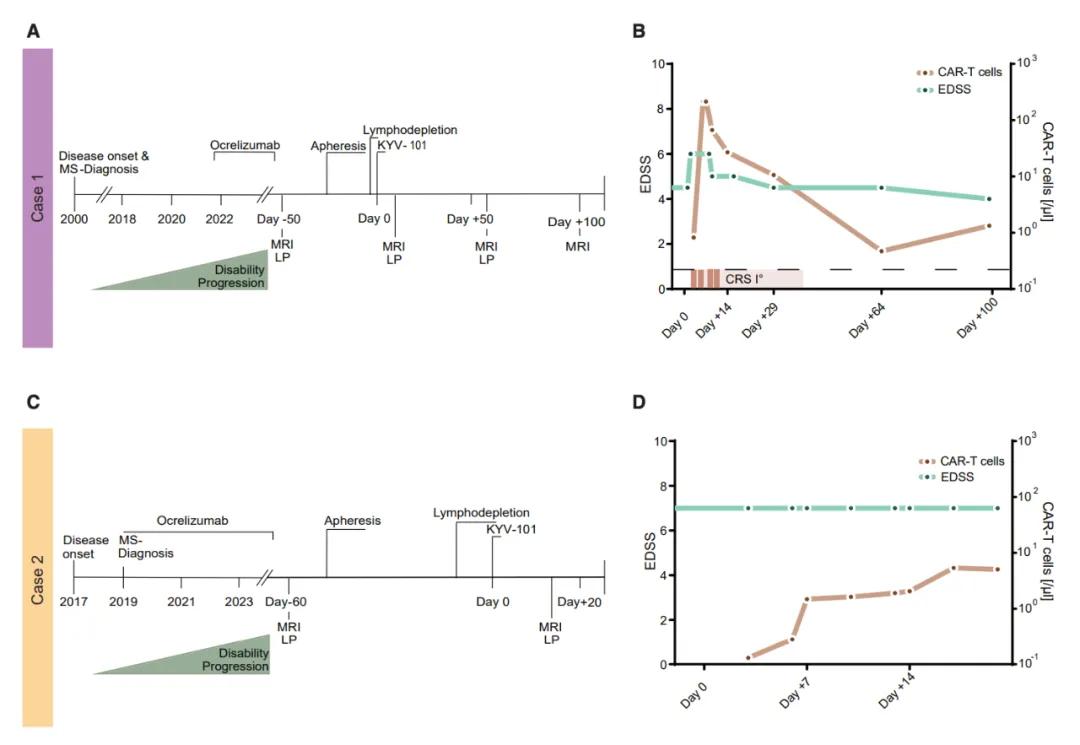
This study enhances the therapeutic potential of CAR T cell therapy in patients with advanced multiple sclerosis that are refractory to conventional antibody-mediated B cell depletion. CD19-targeted CAR T cell therapy not only suppresses recurrence of inflammation, but also removes residual B cells from the central nervous system (CNS) that may lead to disease progression. Further evaluation of the short-and long-term safety and therapeutic efficacy of this therapy are also needed.
Paper link: https: / / www.cell.com/med/fulltext/S2666-6340(24)00114-4
The company's product recommendation
1.376608-71-8 https://www.bicbiotech.com/product_detail.php?id=5369
2.951163-61-4 https://www.bicbiotech.com/product_detail.php?id=5370
3.221221-16-5 https://www.bicbiotech.com/product_detail.php?id=5371
4.327-76-4 https://www.bicbiotech.com/product_detail.php?id=5372
5.98-15-7 https://www.bicbiotech.com/product_detail.php?id=5373




