Targeted protein degradation technology represented by PROTACs has become a promising new strategy for drug development, with more than 30 drug candidates currently in clinical trials. Although PROTACs has achieved the degradation of multiple pathogenic targets, the degradation signals for constructing protein degradation techniques, as well as the type of E3 ligase, are still limited. Currently, only two E3 ligases, VHL and CRBN, are widely used in the design of PROTACs. Therefore, it is urgent to explore other protein degradation signals for constructing new protein degradation technologies. On August 8,2024, Professor Rao Hai's team from Southern University of Science and Technology published a paper titled "Distinct Amino Acid-Based PROTACs Target Oncogenic Kinases for Degradation in Non-Small Cell Lung Cancer (NSCLC)" in Journal of Medicinal Chemistry. The study developed a novel PROTACs (also known as AATacs) based on three amino acids that trigger the N-terminal degradation pathway (proline, glycine and lysine), and successfully degraded two oncogenic protein kinases EML 4-ALK and EGFR-L858R / T790M in non-small cell lung cancer.

N-末端蛋白质降解途径是上世纪80年代发现的一种由泛素介导的蛋白质降解途径,早期的研究确立了位于N-末端的精氨酸和组氨酸等多种氨基酸能够被UBR家族E3连接酶识别,被认为是N-末端蛋白质降解途径的主要信号。最近的一些研究证实了甘氨酸和脯氨酸也可作为N-末端途径的降解信号,其分别能够被CRL2家族的E3连接酶ZYG11B或ZER1以及GID4识别介导蛋白质的降解。因此,为了构建基于N-末端途径的AATacs,作者以Brigatinib为靶蛋白配体,甘氨酸、脯氨酸以及赖氨酸为N-末端蛋白质降解信号,设计了一类靶向降解ALK和EGFR的AATacs。作者首先以脯氨酸为N-末端蛋白质降解信号,探究了合适的Linker长度,结果发现以4个PEG单元为Linker的Pro-PEG3-BA降解效率最优。随后作者固定优势Linker,将脯氨酸替换为甘氨酸和赖氨酸作为N-末端降解信号,设计了一系列AATacs。此外,作者还将脯氨酸替换为无法触发N-末端降解信号的鸟氨酸设计了Orn-PEG3-BA作为阴性对照(图1)。
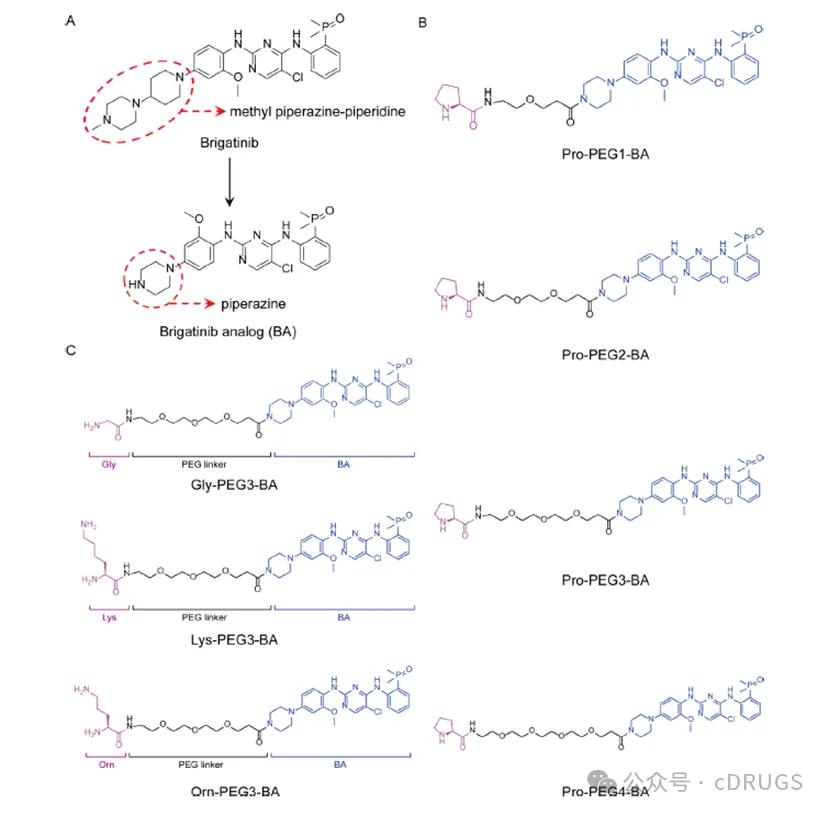
Figure 1 AATacs. Design and structure optimization
Subsequently, the authors explored the degradation efficiency of AATacs from a proline-based constructs containing different PEG, and found that all four compounds could induce pronounced EML 4-ALK degradation in H3122 cells at concentrations greater than 1 μ M. Moreover, this compound also enables degradation of EGFR L858R / T790M in H1975 cells. By comprehensive comparison, the degradation efficiency of Pro-PEG 3-BA is relatively high (Figure 2A). Lys-PEG 3-BA and Gly-PEG 3-BA by replacing proline to lyl and glycine also induced EML 4-ALK degradation in H3122 cells, while no significant EML 4-ALK degradation was observed in the negative control Orn-PEG 3-BA treated group (Figure 2B). The authors also explored the degradation profile of Pro-PEG 3-BA by whole proteomic analysis and showed that ALK levels were the most obvious protein in H3122 cells treated with Pro-PEG 3-BA, which proved the good selectivity of Pro-PEG 3-BA (Figure 2C).
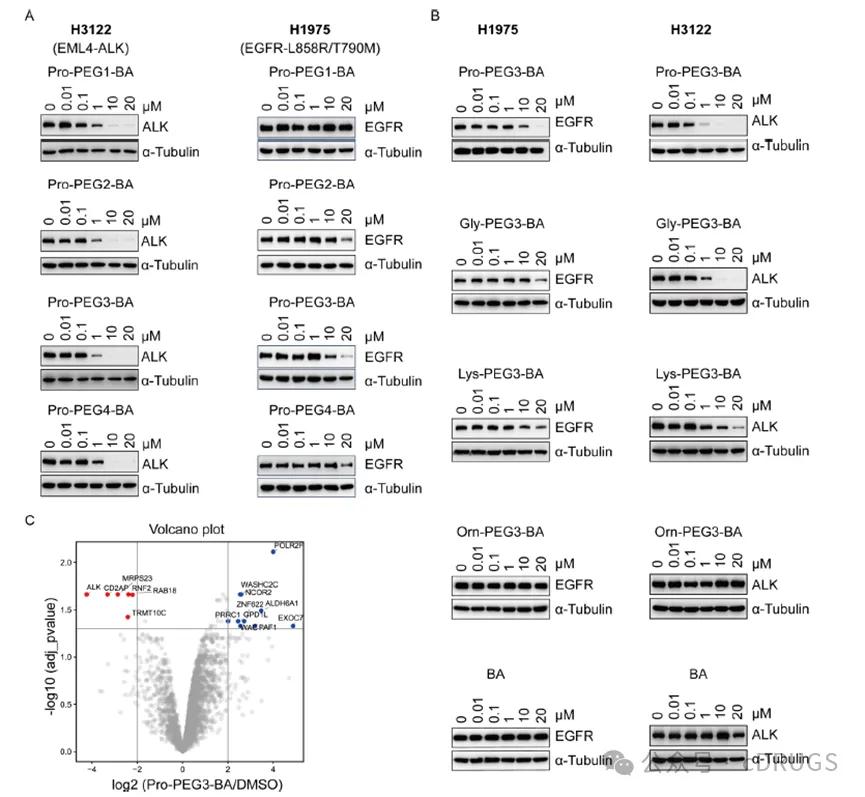
Figure 2 AATacs Assessment of the efficiency and selectivity of EML 4-ALK and EGFR L858R / T790M degradation
Next, the authors further evaluated the DC50 and Dmax of EML 4-ALK and EGFRL858R / T790M based on three amino acid constructs, and showed that Pro-PEG 3-BA, Lys-PEG 3-BA and Gly-PEG 3-BA in H3122 cells were between 0.42 and 1.32 μ M, all able to reach more than 90% of Dmax (Figures 3A and 3B). The three compounds degraded EGFRL858R / T790M relatively poorly, with DC50 between 13.5-20.2 μ M and Dmax between 43 and 69% (Figures 3A and 3B). In addition, the authors also evaluated the value-added inhibition effect of the three compounds on H3122 and H1975 cells by CCK-8. The results showed that the three compounds were IC50 between 0.16-5.30 μ M for H3122 cells and 8.80-20.74 μ M for H1975 cells (Figure 3C).
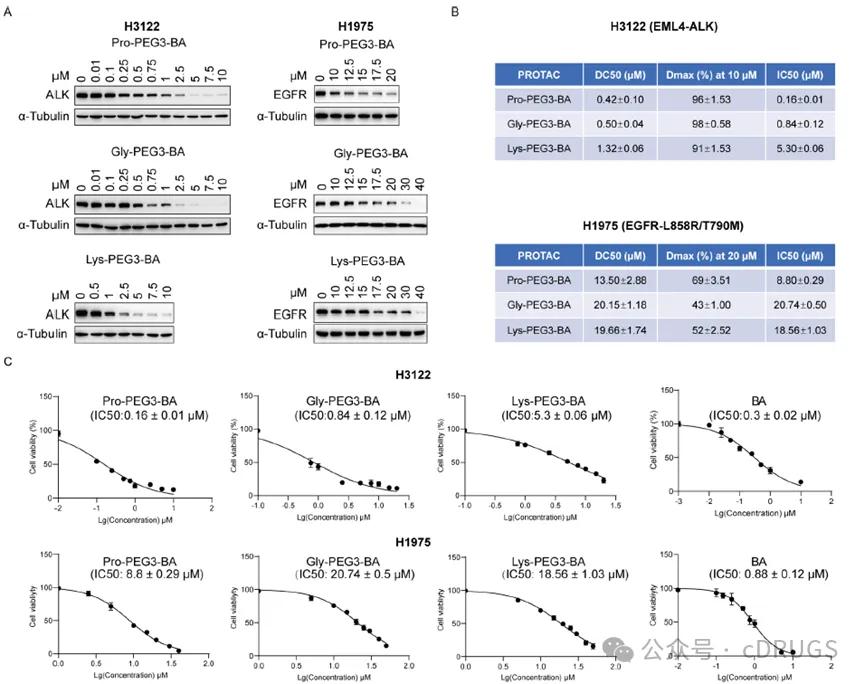
Figure 3 Assessment of degradation efficiency and antiproliferative efficiency of AATacs constructs based on different amino acids
To further explore the mechanism of action of AATAcs-induced degradation of EML 4-ALK and EGFRL858R / T790M, the authors explored the degradation efficiency of Pro-PEG 3-BA in the proteasome inhibitor MG132 and H3122 and H1975 treated with the lysosomal inhibitor chloroquine, respectively. It can be found that MG132 can partially inhibit the degradation of EML 4-ALK and EGFRL858R / T790M by Pro-PEG 3-BA, which indicates that the induction of protein degradation by ATTacs is based on the function of the proteasome (Figure 4A). Furthermore, Pro-PEG 3-BA was able to lead more rapidly to reduced levels of EML 4-ALK and EGFRL858R / T790M in cells treated with Cycloheximide (Figure 4B). Further addition of MG132 was able to inhibit the turnover of EML 4-ALK and EGFRL858R / T790M and prolong its half-life (Figure 4C). Addition of the target protein ligand Brigatinib resulted in an inhibition of EML 4-ALK degradation by Pro-PEG 3-BA (Figure 4D). These results suggest that Pro-PEG 3-BA induces the degradation of the target proteins.Subsequently, to demonstrate whether the induction of protein degradation by AATacs was achieved through the N-terminal degradation pathway, the authors constructed GID 4 knockout H3122 and H1975 cells. It was found that Pro-PEG 3-BA could not degrade EML 4-ALK and EGFRL858R / T790M after GID 4 knockdown (Figures 4E and 4F). Similarly, the degradation capacity of Gly-PEG 3-BA was significantly reduced in the ZYG11B knockdown cells (Figures 4G and 4H). Finally, the authors also evaluated the effects of Pro-PEG 3-BA on cell cycle and apoptosis and showed that Pro-PEG 3-BA was able to induce H3122 and H1975 cell arrest in G1 phase in a concentration-dependent manner and significantly increased the proportion of early and late apoptotic cells.
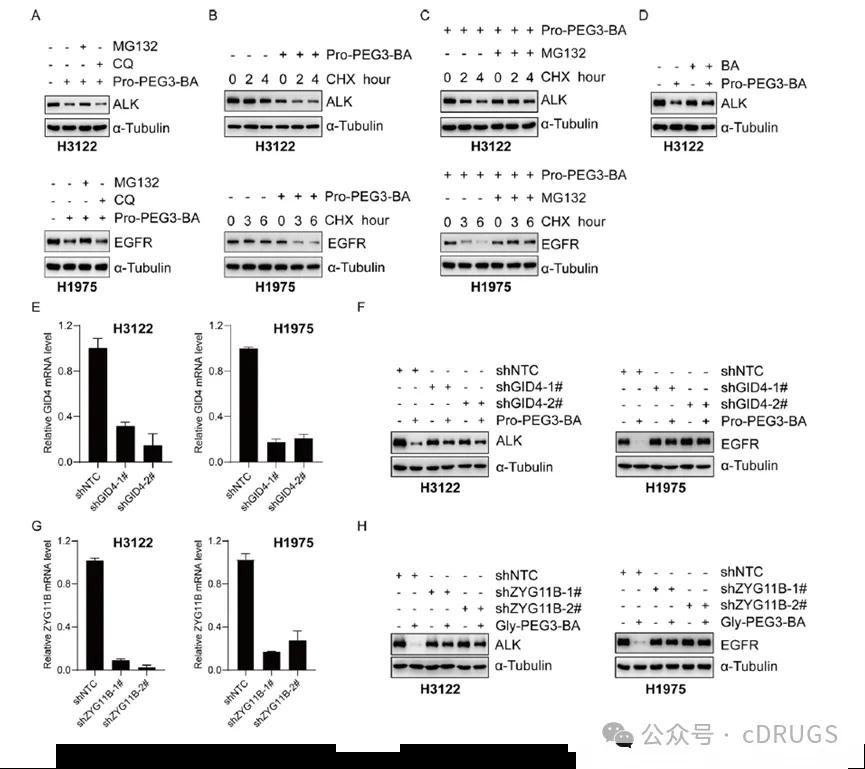
Figure 4. Investigation of the mechanism of action of AATacs
last, The authors also evaluated the PK properties of Pro-PEG 3-BA in a mouse model, The results showed that after intravenous administration of 20 mg / kg, Half-life (T1 / 2) of 2.9 hours, Maximum plasma concentration (Cmax) was 3.7 μ g / mL, Area under the curve (AUC) is 6.0 h * μ g / mL; After the intraperitoneal administration of 50 mg / kg, Half-life (T1 / 2) of 5.2 hours, Maximum plasma concentration (Cmax) of 33 μ g / mL, The area under the curve (AUC) is 81 h * μ g / mL, The bioavailability was 53.2% (Figures 5A and 5B). PD experiments in the H132 xenograft model showed that Pro-PEG 3-BA significantly reduced ALK protein levels in tumor tissue at 10 mg / kg by intraperitoneal injection (Figure 5C).
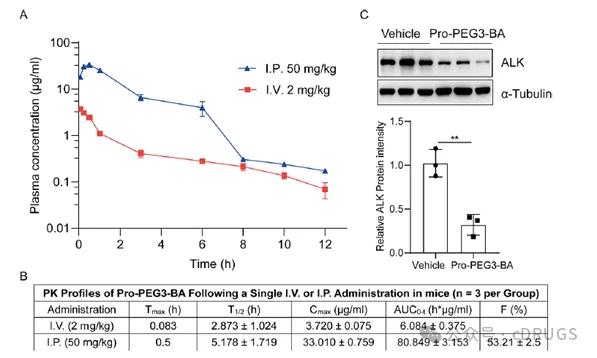
Figure Figure 55 The PK and PD studies of Pro-PEG 3-BA
In conclusion, this study used proline, glycine and lysine as degradation signals to successfully construct a class of AATacs based on N-terminal degradation pathway, systematically explored the degradation efficiency and degradation mechanism of oncoproteins EML 4-ALK and EGFRL858R / T790M in tumor cells, and preliminarily evaluated its therapeutic potential for NSCLC. This study expands the E3 ligase and protein degradation signal types in the limited field of protein degradation, and provides a new idea for the construction of new protein degradation technologies.
Link: https: / / doi.org/10.1021/acs.jmedchem.4c00208
The company's product recommendation:
1.1683-85-8 https://www.bicbiotech.com/product_detail.php?id=5395
2.23056-35-1 https://www.bicbiotech.com/product_detail.php?id=5396
3.313-12-2 https://www.bicbiotech.com/product_detail.php?id=5397
4.39226-97-6 https://www.bicbiotech.com/product_detail.php?id=5398
5.106261-48-7 https://www.bicbiotech.com/product_detail.php?id=5399




