B cell development begins in the bone marrow with B cell lineage differentiation by lymphoid precursor cells
From early pro-B, the cells transition to Pre-B cells.
B cells rearrange the heavy and light chain fragments encoded by their genes to form a unique single-reactive (mono-reactive) BCR.
In the bone marrow and further differentiated into immature (immature) B cells, the B cells express the first BCR.
To ensure self-tolerance, self-reactive immature B cells undergo receptor editing or are eliminated by negative selection in the bone marrow (central tolerance) and secondary lymphoid organs (peripheral tolerance);
Surviving B cells without self-response, in the form of transitional (transitional) B cells, are released from the bone marrow and mature in the spleen. Outside of the bone marrow, B cell maturation can be divided into two types: TLR-mediated extra-follicular (extra-follicular) B cell maturation and follicular B cell maturation requiring T cells. The former does not necessarily require T cells to generate plasmablasts and short-lived plasma cells; the latter requires T cells, involving the affinity maturation (affinity maturation) of B cells in lymphoid follicles and leading to the generation of memory B cells and long-lived plasma cells that re-enter the bone marrow and remain in a specific location (niches) for a long time.
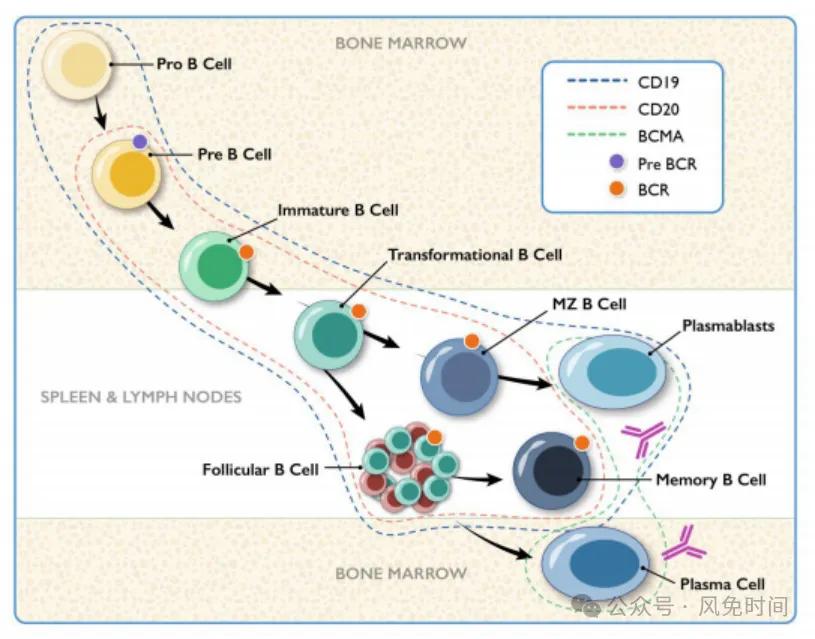
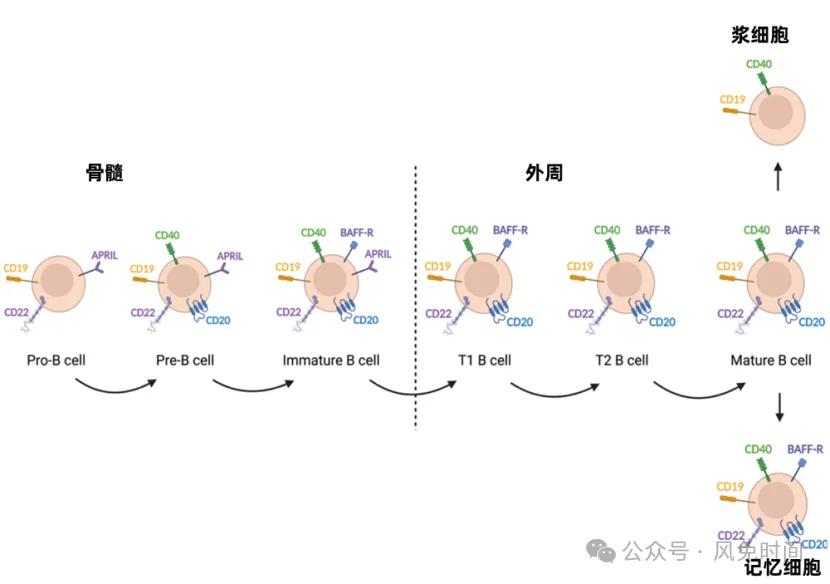
Surface markers expressed at different stages of B cell maturation, including CD19, CD20, CD22, CD40, etc
SLE is an autoimmune disease developed by the interaction of genetic factors and environmental triggers, characterized by aberrant self-responses to DNA and nuclear proteins as well as type I interferon responses.
B cell-mediated autoantibody production, is thought to be a critical step in the occurrence of SLE immunopathology.
In the pathology of systemic lupus erythematosus (SLE), the mechanism of central B cell tolerance or peripheral B cells in secondary lymphoid organs is changed, forming autoreactive B cells and escape.
Both extrafollicular and intrafollicular B cell maturation, may play a role in the formation of autoreactive B cells, leading to the production of autoantibodies against dsDNA and other nuclear proteins by plasmablasts, short-lived plasma cells, and long-lived plasma cells of SLE patients.
The abnormal balance of peripheral B cell subsets, including regulatory B cell dysfunction, increased T-bet + CD11c + B cells, and plasmablasts, is strongly associated with disease activity, autoantibody production, and organ damage in SLE,
CD20 is the main target antigen for B cell clearing therapy, which is expressed in a broad B cell lineage from pre-B, cells to memory B cells, but not in plasmablasts and plasma cells.
CD19 is another B cell specific receptor that covers a wider range of B cell lineage and also includes pro-B and plasma cells, but is not expressed on long-lived plasma cells.
Therapeutic strategies targeting B cells include direct killing of B cells, regulation of B cell function, inhibition of molecules essential for B cell growth and survival, and acceleration of autoantibody clearance, etc. The current main strategies are as follows:
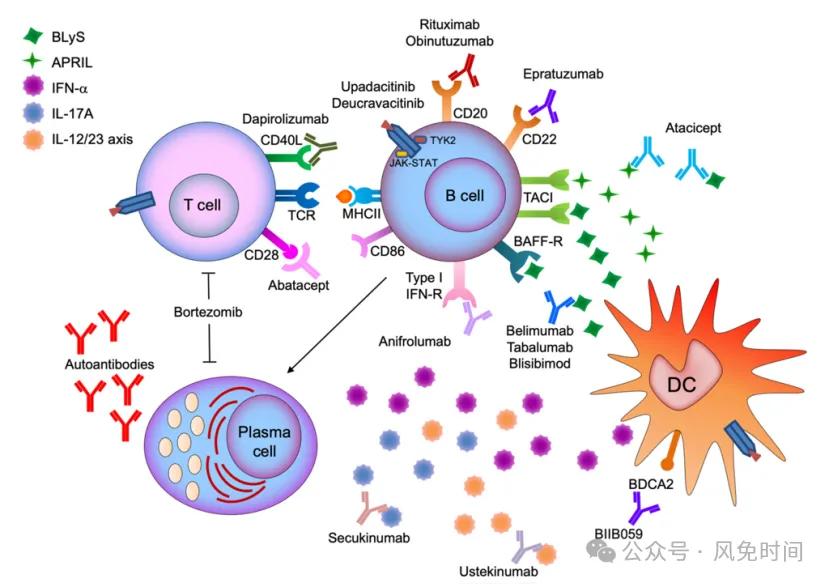
Anti-CD19 obexelimab (phase 2 clinical study not reaching the endpoint) and CAR-T (single case report);
Anti-CD20 rituximab (phase 3 clinical study did not reach the end point, but can reduce anti-dsDNA antibody, proteinuria, improve complement), Obbituzumab (obinutuzumab, OBZ), aurelizumab (phase 2 did not reach the end point, but some renal reaction), etc.;
Beliuumab against B cell activation factor (BAFF) (decreased disease activity and hormone dose), tabalumab (stage 3 did not reach the end point), etc.;
BAFF / APRIL resistance (stage 2b reaches the primary endpoint), etc.;
Targeting type I interferons (IFN-I): IFN- α is produced by B cells and plasmacytoid dendritic cells that stimulates B cell differentiation and promotes autoantibody production through BAFF production. Anifrolumab is a fully humanized monoclonal antibody to IgG1K that binds to IFNAR and inhibits pro-inflammatory cytokines by blocking the IFN-I self-amplifying ring.
Targeting the CD40 ligand (CD40L): CD40L on the surface of T cells interacts with CD40 on B cells and is essential for the growth, differentiation, and activation of B cells. Both monoclonal antibodies Ruplizumab and Toralizumab against CD40L were terminated by thrombotic events in SLE studies. A pegylated anti-CD40LFab fragment, Dapirolizumab pegol, whose absence of Fc fragments and inability to bind platelet receptors, reduces the risk of platelet aggregation. Dapirolizumab Phase clinical trials of efficacy and safety in patients with moderate to severe SLE are ongoing, and its potential to treat SLE needs to be further defined.
BTK inhibitors and proteasome inhibitors, etc.
The company's product recommendation:
1.180181-93-5 https://www.bicbiotech.com/product_detail.php?id=5435
2.269078-77-5 https://www.bicbiotech.com/product_detail.php?id=5436
3.269078-79-7 https://www.bicbiotech.com/product_detail.php?id=5437
4.269078-81-1 https://www.bicbiotech.com/product_detail.php?id=5438
5.269078-76-4 https://www.bicbiotech.com/product_detail.php?id=5439




