Bone organoids as pioneering tools in biomedical research bridge the gap between theoretical knowledge and practical applications and help us in our understanding of bone biology and related diseases. These tiny, self-organizing structures mimic the complexity of bone tissue, providing a dynamic platform for scientific discovery and medical innovation.
The rapidly evolving bone organoid technology provides new models for the research and clinical applications of bone-related diseases. With the continuous optimization of technology and the integration of multiple disciplines, its application prospect will be further expanded.
Organoids and their development history
Organoids (organoids) is a small, organoid cell mass formed in a three-dimensional culture system that can imitate the structure and function of organs in the body. They have the following remarkable characteristics: ① Multiple cell types: organoids are composed of a variety of different types of cells that interact to form a complex three-dimensional structure. This multicellular composition enables the organoids to more accurately mimic the microenvironment and physiological functions of the organs in vivo.② Self-organization: Under specific culture conditions, organoids can self-organize and differentiate, forming a structure similar to the organs in the body. This self-organizing ability makes organoids an important model for the study of cell behavior and tissue development.③ Functional: Organoids have certain functions of the organs in vivo, such as metabolism, secretion, and the ability to respond to external stimuli. Organoid technology has demonstrated wide application potential in the biomedical field and has become an important tool in basic research, disease models, drug screening and regenerative medicine.
Scientists have successfully produced a variety of different types of organoids, including intestinal organoids, brain organoids, liver organoids, kidney organoids and other [1]. These organoids are highly structurally and functionally mimic the in vivo organs, providing more realistic models for biomedical research. Organoid techniques are widely used to establish disease models and to perform drug screening. This technology involves leveraging patient-derived cells to produce personalized organoid models that can highly simulate the pathological processes of specific diseases. By extracting cells from the patient, the scientists were able to grow organoids that were highly consistent with the patients condition. These organoids retain the patient genetic information and pathological features that can be used to deeply study the disease onset and progression of [2].
Personalized organoid models have important applications in anticancer drug screening. Scientists can use these models to test the effects of various anticancer drugs, thus choosing the most effective personalized treatment options. By testing different chemotherapeutic drugs in patient-derived intestinal organoids, the investigators can determine which drug is most lethal to cancer cells in a given patient. This personalized drug screening approach greatly improves the accuracy and effectiveness of treatment and reduces side effects [3]. Organoid technology also holds important applications in regenerative medicine. Scientists hope to repair damaged organ tissue through organoid technology, and even achieve organ transplantation in the future. As "seed cells" in tissue engineering, organoids are expected to be used to construct functional tissues or organs that, through in vitro culture and engineering techniques, generate biological tissues or organs that can be used for transplantation. For example, by combining organoids with biological scaffold materials, scientists can build biologically active three-dimensional tissue structures to repair or replace damaged tissue [4].
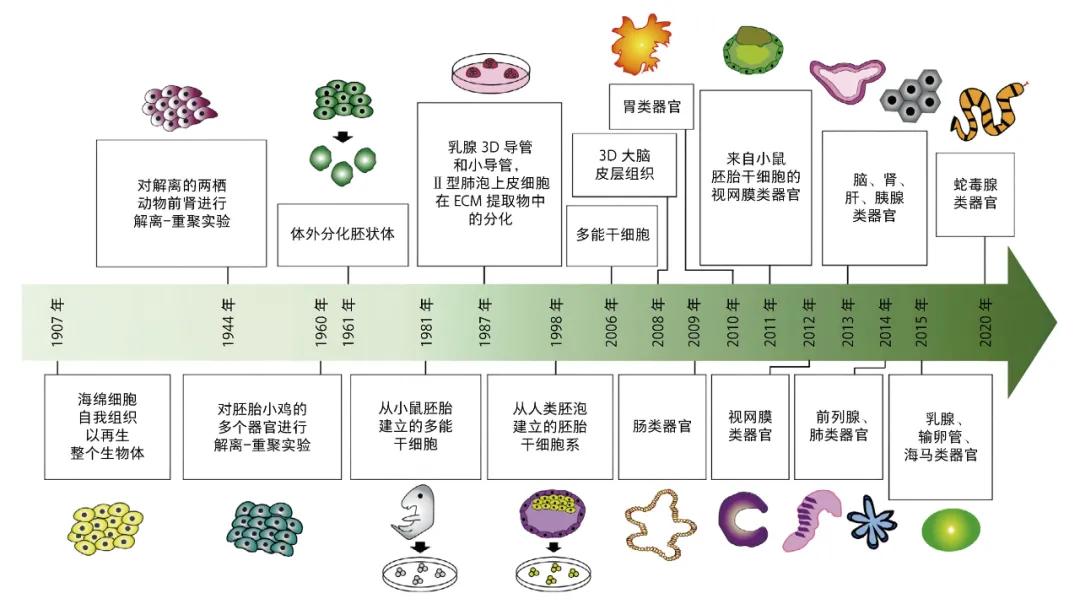
Timeline for the development of the organoid cultures [6]
The development of organoid technology has undergone a long and tortuous process, with concepts and methods dating back to the early 20th century.
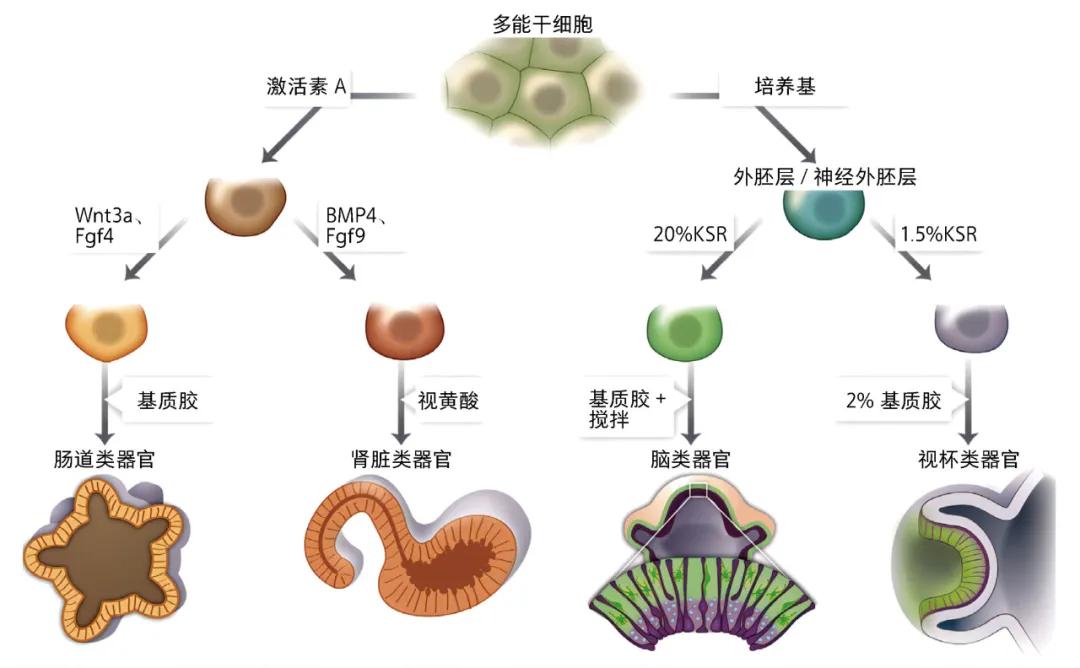
[5] Wnt 3 a: apless MMTV integration site family member 3A; Fgf4: fibroblast growth factor 4; BMP 4: bone morphogenetic protein 4; Fgf9: fibroblast growth factor 9; KSR: knockout serum replacement.
Early exploration (early 20th century)
In the early 20th century, scientists began trying to grow tissues and organs in the laboratory. These early experiments included in vitro cultured tissue blocks of embryonic organs. These attempts laid the foundation for later cell culture techniques, and although the technical means and theoretical knowledge were still very limited at that time, these explorations accumulated valuable experience for later research.
2-dimensional culture (1950s)
By the 1950s, important progress was made in cell culture techniques. In particular, the development of two-dimensional cell culture technology has enabled scientists to grow monolayers of cells in culture dishes. This advance not only greatly improves the controllability and repeatability of the experiment, but also lays the foundation for further three-dimensional cultivation. The wide application of two-dimensional culture technology has led to the rapid development of cell biology research and created conditions for the breakthrough of organoid technology.
3-dimensional culture (1990s)
In the 1990s, scientists began to explore three-dimensional cell culture techniques to better simulate the cell environment in the body. American biologist Bissell (M. Bissell) and his colleagues used three-dimensional matrix to cultivate mammary epithelial cells and successfully simulated the formation of mammary acini [7]. This breakthrough study demonstrates the potential of 3 D culture techniques to simulate complex tissue structures and points the way for the development of organoid technology.
Breakthrough in organoid technology (early 2000s)
The real technological breakthrough came in 2009, when the team of H. Clevers from Utrecht) in the Netherlands first successfully cultured [8] of intestinal organoids in vitro using adult stem cells. This advance marks the formal birth of organoid technology, making it an important tool for disease research, drug screening, and regenerative medicine. The teams research proves that organoids can not only be cultured in the laboratory, but also maintain their complex structure and function in the body. This discovery has stimulated a strong interest and enthusiasm for organoid research among scientists worldwide.
Exploration of bone organoids
Bone organoids (bone organoids) refers to the three-dimensional self-renewal and self-organized micro-bone tissue constructed in vitro by biologically active materials and directed differentiated stem cells (such as backbone cells, embryonic stem cells, etc.) or progenitor cells (such as osteoblasts and / or osteoclasts, etc.). These bone organoids have the spatial characteristics of biomimulation and are able to recapitulate the complex biological functions of bone tissue in vivo in a three-dimensional culture system. Compared with traditional two-dimensional cell culture systems and animal experiments, bone organoids can better mimic the natural physiological microenvironment of bone, providing a more physiologically relevant model for in vitro research [9]. Bone organoids are an emerging field of organoid technology dedicated to model the formation and function of bone tissue in vitro. Unlike other types of organoids, bone organoids are their specific and challenging. These particularities and challenges are not only derived from the complex structure and diverse functions of the bone tissue, but also involve its unique developmental processes and mechanical environment.
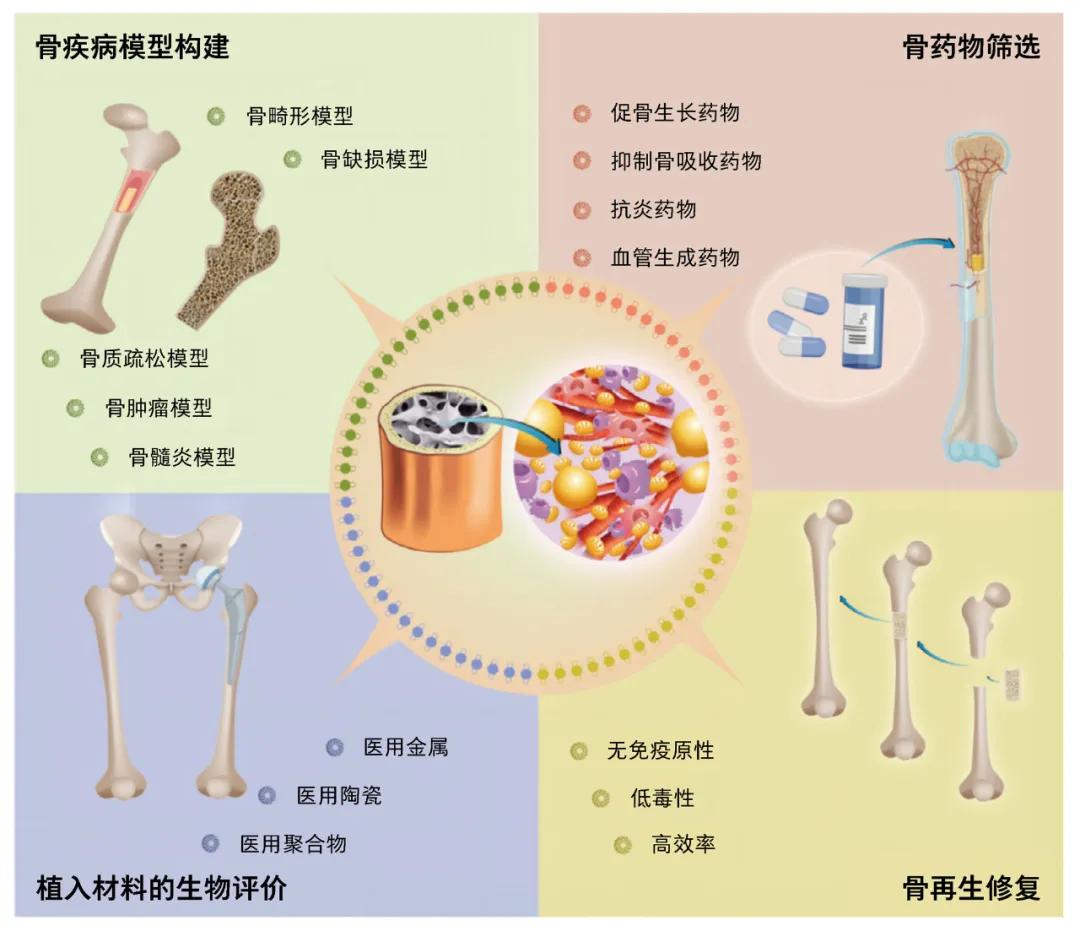
Application prospects for bone organoids [9]
The particularity of bone organoid construction
Bone organoids are specific in culture. First, bone tissue bears mechanical stresses in vivo, which are critical for bone formation and remodeling. Thus, cultivating bone organoids requires simulation of similar mechanical environments, often by application of mechanical stress or utilization of bioreactors, different from other organoids that rely primarily on chemical and biological signals. Secondly, the mineralization process of bone tissue is an important feature. The matrix secreted by osteoblasts gradually calcifies to form hard bone. Therefore, the cultured bone organoids need to add appropriate mineralization inducer to the culture medium to promote the mineralization of osteoblasts, while most other organoids do not involve the formation of such hard tissue. Again, the formation of bone organoids not only depends on osteoblasts, also need the participation of osteoclasts and other mesenchymal cells, scientists must design co-culture system, make different cell types interaction, complete the formation and maintenance of bone tissue, the complexity of the multicellular co-culture system makes the study has special challenges. Finally, bone tissue has rich vascular network, simulate the vascularization is crucial for cultivating functional bone organoids, researchers usually introduce vascular endothelial cells or using tissue engineering technology, promote the formation of blood vessels in organoids, in order to improve the physiological authenticity and functionality of organoids, this is different from most other organoids, the latter usually does not need to simulate complex vascular network.
Challenging construction of bone organoids
The cultivation of bone organoids faces numerous challenges arising mainly from their complex tissue architecture and specific physiological needs. First, bone tissue includes not only the mineralized matrix but also the bone marrow cavity and various cell types, and successful cultivation of this complex structure requires fine control and optimization of culture conditions, far more complex than cultivating organoids with a single cell type or simple structure. Secondly, bone tissue is constantly affected by dynamic mechanical stresses in vivo, such as compression, stretching and shear forces, which not only affect the function of bone cells, but also play a key role in the formation and remodeling of bone matrix. Therefore, for the in vitro culture of bone organoids, devices need to be designed that can simulate these dynamic mechanical environments to ensure that the cultured organoids have real physiological functions. Again, cultured bone organoids requires specific growth factors and nutrients to promote osteoblast differentiation and mineralization, which requires precise regulation of medium components to mimic the body microenvironment, which is different from the culture of other organoids, the latter usually does not need such a precise and diversified growth factor regulation. Finally, the formation and maturation of bone tissue takes long times, and also requires long culture periods when cultivating bone organoids in vitro, which increases the uncertainty and challenges during culture, such as how to prevent cell senescence and death during culture, and how to keep the culture environment stable.
Bone organoids version 1.0
The authors team used the "one-pot method" to synthesize a new bioink [10] for bone tissue engineering, and used 3D bioprinted bone stent to build a [11] of large-size bone organoids that can be used in time culture, multicellular differentiation and self-mineralization, and applied it to the bone regeneration study of large bone defects. Team staff through the design of new suitable bone tissue engineering biological ink, using digital light processing biological printing technology, accurate copy bone tissue complex microscopic structure, and then in the body for the long time directional cultivation, make differentiation into a variety of bone marrow cells (blood cells, immune cells, vascular endothelial cells, chondrocytes, osteoblasts, fat cells and osteoclasts), good mechanical properties (youngs modulus class, close to cancellous bone), space topology class bone (loose porous micro-nano structure) and large size (cm) functional bone organoids. Its application to the regenerative repair of the bone defect model showed a strong repair capability. Bone organoids version 1.0 can be used to establish bone disease models, screen drugs for bone metabolism, and perform biological evaluation of bone implant material. Imagine, in the future, for patients who have bone defects or necrosis for various reasons, it may be possible to extract their stem cells to build bone organoids and then implant them into specific parts of the body, such as the abdomen. It can be expected that these constructs will grow and mature into true bone tissue that can then be used for bone grafting in areas affected by defects or necrosis.
Conclusion and Outlook
As an important branch of organoid research, bone organoid technology has shown great potential in regenerative medicine, bone disease research and drug screening, despite many challenges such as complex tissue structure, dynamic mechanical environment simulation, precise regulation of growth factors and nutrients, and prolonged culture. Through continuous technological innovation and optimization, scientists have made remarkable progress and successfully developed bone organoid models with physiological function. In the future, bone organoid technology will combine with gene editing, 3D biological printing and intelligent materials, the combination of standardization and scale culture methods, optimization of mechanical environment simulation technology, and strengthen the combination of basic research and clinical application, promote its in bone regeneration and repair, bone disease treatment of clinical transformation. The development of bone organoid technology has a broad prospect. With the continuous progress of technology, it is expected to bring revolutionary changes to the diagnosis, treatment and regenerative medicine of bone tissue related diseases in the future, benefit more patients, promote the development of biomedical field, and become an important tool to solve the medical problems related to bone tissue.
Jian Wang: PhD student, Institute of Translational Medicine, Shanghai University, Shanghai 200444.
Su: Professor, Institute of Translational Medicine, Shanghai University; Chief Physician, Xinhua Hospital Affiliated to School of Medicine, Shanghai Jiao Tong University, Shanghai 200092.
Wang Jian: Doctoral Candidate, Institute of Translational Medicine, Shanghai University, Shanghai 200444.
Su Jiacan: Professor, Institute of Translational Medicine, Shanghai University, Shanghai 200444; Chief Physician, Xin Hua Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200092.
References:
[1]Xu H, Jiao Y, Qin S, et al. Organoid technology in disease modelling, drug development, personalized treatment and regeneration medicine. Experimental Hematology & Oncology, 2018, 7: 30.
[2]Chitrangi S, Vaity P, Jamdar A, et al. Patient-derived organoids for precision oncology: A platform to facilitate clinical decision making. BMC Cancer, 2023, 23: 689.
[3]Yu Y Y, Zhu Y J, Xiao Z Z, et al. The pivotal application of patient-derived organoid biobanks for personalized treatment of gastrointestinal cancers. Biomarker Research, 2022, 10: 73.
[4]Liu N, Zhang X, Guo Q, et al. 3D bioprinted scaffolds for tissue repair and regeneration. Frontiers in Materials, 2022, 9.
[5]Lancaster M A, Knoblich J A. Organogenesis in a dish: Modeling development and disease using organoid technologies. Science, 2014, 345: 1247125.
[6]Corrò C, Novellasdemunt L, Li V S W. A brief history of organoids. American Journal of Physiology Cell physiology, 2020, 319: C151-C165.
[7]Bissell M J, Rizki A, Mian S. Tissue architecture: The ultimate regulator of breast epithelial function. Current Opinion in Cell Biology, 2003, 15: 753.
[8]Sato T, Vries R G, Snippert H J, et al. Single lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature, 2009, 459: 262-265.
[9]Chen S, Chen X, Geng Z, et al. The horizon of bone organoid: A perspective on construction and application. Bioactive materials, 2022, 18: 15-25.
[10]Ren X, Wang J, Wu Y, et al. One-pot synthesis of hydroxyapatite hybrid bioinks for digital light processing 3D printing in bone regeneration. Journal of Materials Science & Technology, 2024, 188: 84-97.
[11]Wang J, Wu Y, Li G, et al. Engineering large-scale self-mineralizing bone organoids with bone matrix-inspired hydroxyapatite hybrid bioinks. Advanced materials (Deerfield Beach, Fla), 2024, e2309875.
This article is published in Volume 76,2024, Science, Issue 4 (P23-P26)
Key words: bone organoid bone tissue engineering 3D bioprinting regenerative medicine
The company's product recommendation:
1.117391-54-5 https://www.bicbiotech.com/product_detail.php?id=5445
2.117391-48-7 https://www.bicbiotech.com/product_detail.php?id=5446
3.117391-57-8 https://www.bicbiotech.com/product_detail.php?id=5447
4.115957-22-7 https://www.bicbiotech.com/product_detail.php?id=5448
5.117391-52-3 https://www.bicbiotech.com/product_detail.php?id=5449




