
abstract
Background and Objective: In the past few years, it is recommended to demonstrate the bioequivalence (BE) of several topical skin drugs. An important component of this approach is the comprehensive characterization of the physical and structural properties (Q3) of complex topical semisolids. The purpose of this study is to determine whether the topical lidocoine and procaine Q3 characterization contrasts can be used to predict comparative product properties to evaluate the skin pharmacokinetics (PK) of lidocoine and prodocaine in vitro and in vivo.
Methods: The products evaluated in this study included (1) reference EMLA ® (lidoccoine) cream (2.5%: 2.5%); (2) EMLA ® cream and (3) Oraqix® (lidocaine: protracine) gel (2.5%: 2.5%) compound at the same size as lidocaine / protracine cream. The Q3 properties of the three drugs were evaluated. The skin pharmacokinetics (PK) of lidocaine and procaine in gel and cream were compared by in vitro permeation assay (IVPT). BE of cream generics, Oraqix® gel, and EMLA ® cream was assessed according to the skin PK endpoints for lidocaine and prominidocaine. The skin bioavailability of EMLA ® and Oraqix® was also compared for in vivo studies using dermal open flow microperfusion (dOFM) in six healthy subjects.
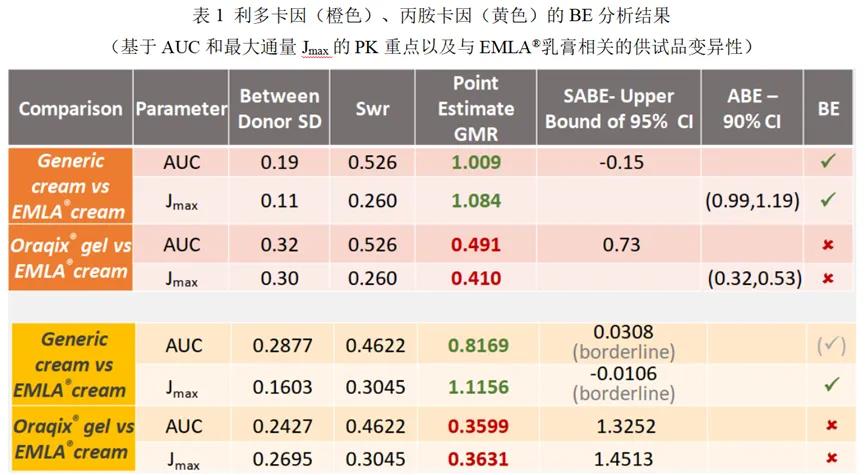
Results: The Q3 characteristics of the reference preparations and generic lidocaine / prominocaine topical cream were similar to each other, while the Oraqix® gel had lower pH, higher evaporation rate, lower yield stress and no droplets compared to the cream product. The results of the IVPT study showed that the skin PK of lidocaine and procaine was comparable between the reference preparation and the generic cream. Lower maximum flux (Jmax) and area under the curve (AUC) of lidocine in Oraqix® gel compared to EMLA ® cream, and the gel and cream were found to be bioequivalent. The results of the in vivo skin PK studies using dOFM in healthy subjects were consistent with the in vitro (IVPT) results.
Conclusion: The results demonstrate a correlation between Q3 similarity of three topical agents or other product properties (skin PK), either in vitro (IVPT) or in vivo (dOFM). The similarity in Q3 properties between reference cream and generic cream correlated accurately and predictable with the comparable bioavailability (and bioequivalence) of lidocaine and propylene aine, while the differences in Q3 properties between reference cream and gel correlated accurately and predictable with differences in bioavailability.
introduce
In the past few years, group weighting has been recommended to support the demonstration of the bioequivalence of multiple topical drugs. Another important component of this approach is a comprehensive characterization of the physical structural properties of complex external semi-solid agents (Q3).
The purpose of this study is to determine whether the comparison of topical topical lidocaine / propilocaine formulation Q3 properties can be used to predict comparative product properties, as assessed by comparing the in vitro and in vivo skin pharmacokinetics (PK) of lidocaine and propilocaine. The specific objectives of this study will include:
Characterize and compare the Q3 properties of cream and gel preparations, each containing lidocaine and procaine; and gel properties using the in vitro and in vivo skin PK studies.
Materials and Methods
The products evaluated in this study included (1) reference preparation EMLA ® (lidocaine; propocaine) topical cream (2.5%; 2.5%), (2) EMLA ® cream and (3) Oraqix® (lidocaine) dental gel (2.5%; 2.5%) with the same specifications. These 3 preparations evaluated Q3 properties including microscopic examination, pH, evaporation rate, and rheological behavior. The skin PK of lidocaine and proacine in the gel and cream was compared by in vitro permeability (IVPT), designed as a repeat study (6 skin donors, 6 samples per donor) using a thermally separated human epidermis and circulation diffusion system. Bioequivalence of generic cream and Oraqix® gels with EMLA ® cream was assessed based on the skin PK endpoints of lidocaine and propylene aine using reference standardized mean bioequivalence (SABE) analysis and 90% confidence interval (CI) assessment. EMLA ® and Oraqix® were also compared by open skin flow microperfusion (dOFM) in another in vivo study in six healthy subjects. The dose for all products in the IVPT and dOFM studies was 10mg / cm2.


results and discussion
Quality detection and Q3 properties
Lidocaine / propanidincaine cream reference and generic Q3 have similar properties but are different from Oraqix® gel:
The mean pH for lidocaine / prominocaine cream reference, generic and Oraqix® gel were 9.10,8.90 (i. e. 9.0 ± 0.1) and 7.65, respectively.
The cream microscopy images show a droplet size diameter of 1 – 3 μ m, while the Oraqix® gel is a uniformly emulsion-less system. Under cryo-scanning electron microscopy (cryo-SEM), the cream exhibited a different microstructure from the gel (Figure 2).
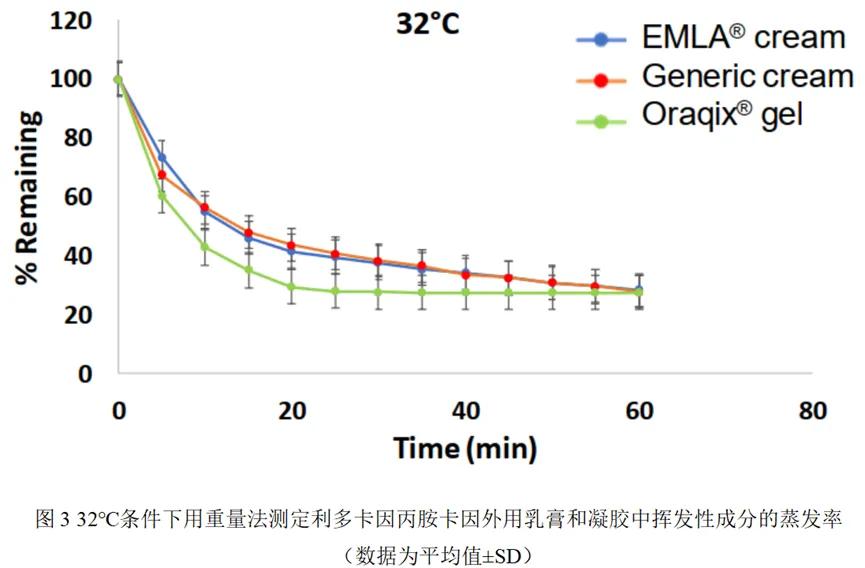
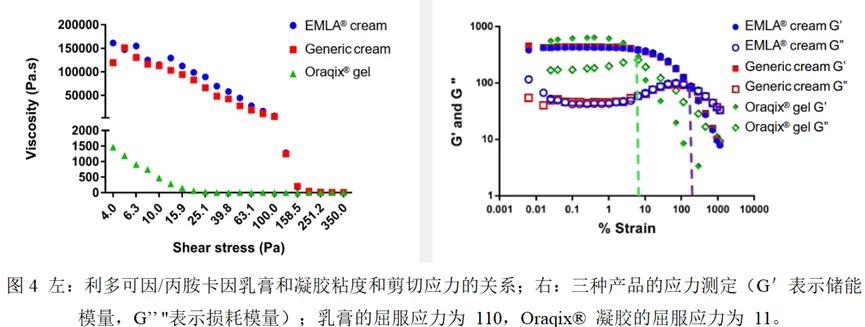
Properties determination
1.for in vitro skin PK studies using IVPT
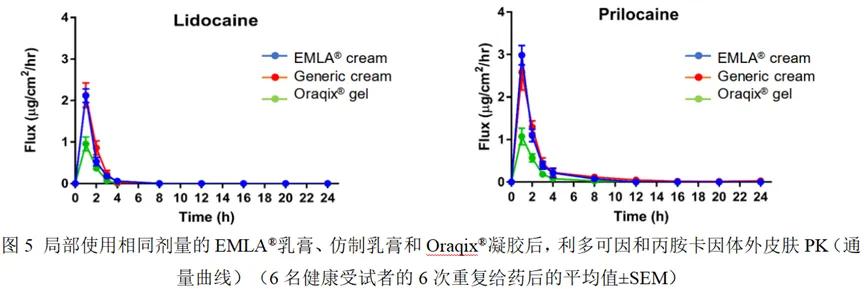
2.for in vivo skin PK studies using dOFM
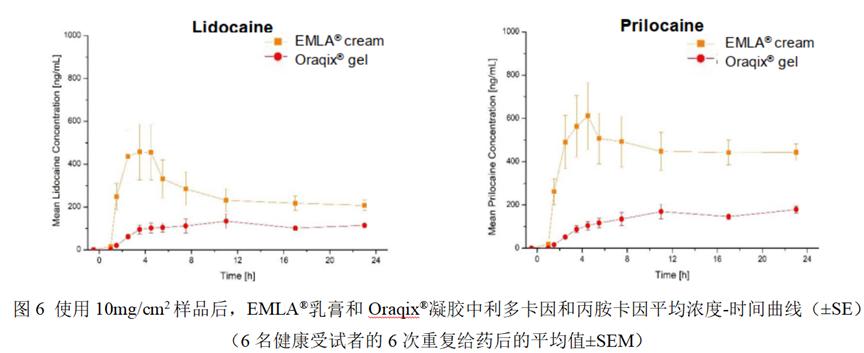
conclusion
The results demonstrate a correlation between the Q3 similarity (or difference) of the three products and their corresponding skin PK, including in vitro (IVPT) and in vivo (dOFM). The Q3 property similarity between the reference cream and the generic cream correlated accurately with the comparable bioavailability (and bioequivalent) similarity of lidocaine and protracine between the two creams and predicted bioequivalence between the two creams. Differences in Q3 properties between the reference preparation cream and gel preparations are accurately correlated with differences in bioavailability and can be predicted.
Our product recommendation:
1.221636-18-6 https://www.bicbiotech.com/product_detail.php?id=5480
2.59855-69-5 https://www.bicbiotech.com/product_detail.php?id=5481
3.71690-89-6 https://www.bicbiotech.com/product_detail.php?id=5482
4.1029234-11-4 https://www.bicbiotech.com/product_detail.php?id=5483
5.1171478-64-0 https://www.bicbiotech.com/product_detail.php?id=5484




