Medical diagnostic technology plays a very important role in the diagnosis, treatment and prevention of diseases. With the continuous development of disease-related biomarker detection strategies, DNA-based biosensors have received increasing attention in clinical diagnostics because of their ability to target genetic information in humans, viruses and bacteria. At the same time, point-of-care diagnosis (POCT) technology has gained significant attention over the past few years because of its potential to improve patient treatment outcomes (e. g., reducing medical costs, reducing diagnostic cycles, and improving access to medical services).
However, although the existing POCT DNA biosensors have good application prospects, they still have some limitations. First, DNA detection using existing DNA biosensors involves multiple steps, such as sample preparation, amplification, and detection, and these steps are performed separately. Therefore, on the one hand, the complexity of the operation requires trained personnel; on the other hand, the spatial requirements for the system to operate effectively challenge its accessibility, especially in the context of field application. More importantly, the cost factor remains a major obstacle due to the need for expensive reagents and materials during the application.
The key to advancing the development of next-generation DNA biosensors is to transform the diagnostic system from a lab to a miniaturization platform by using on-on-chip lab (lab-on-chip) technology. This transformative approach enables a variety of laboratory functions through miniaturization, simplification, and automated strategies.
Microfluidic is a technique designed to manipulate fluid within micron-scale features. The microfluidic platform enables to handle complex analytical processes with smaller solution volumes and fewer infrastructure requirements than laboratory-based operating platforms. However, most microfluidic devices rely on an external power pump to drive solution flow within the microchannel, thereby reducing its portability and hindering its application in POCT. Therefore, the development trend of microfluidic devices with integrated DNA biosensors is that the materials are cheap, easy to operate, and do not rely on external power units.
According to Mmes Consulting, in the near future, Researchers from Seon University (Sogang University) in Korea published a review article in Advanced NanoBiomed Research entitled "Point-of-Care Testing (POCT) Devices for DNA Detection: A Comprehensive Review", An overview of the development of chip laboratory technologies for integrated DNA biosensors in the POCT field.
In this review, the authors first describe the types of microfluidic devices used in DNA detection; then explore the influence of probe selection in DNA microfluidic chip; then review different DNA detection techniques and their applications in DNA microfluidic chip systems. Finally, the author review the current development of DNA microfluidic chips and discuss their application direction in POCT, aiming to narrow the gap between research and practical application.
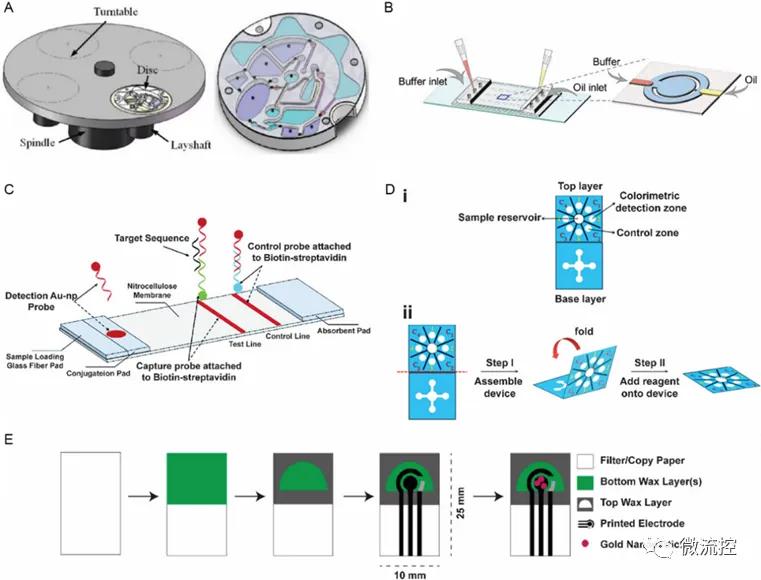
Point-of-care diagnosis (POCT) microfluidic platform for DNA testing
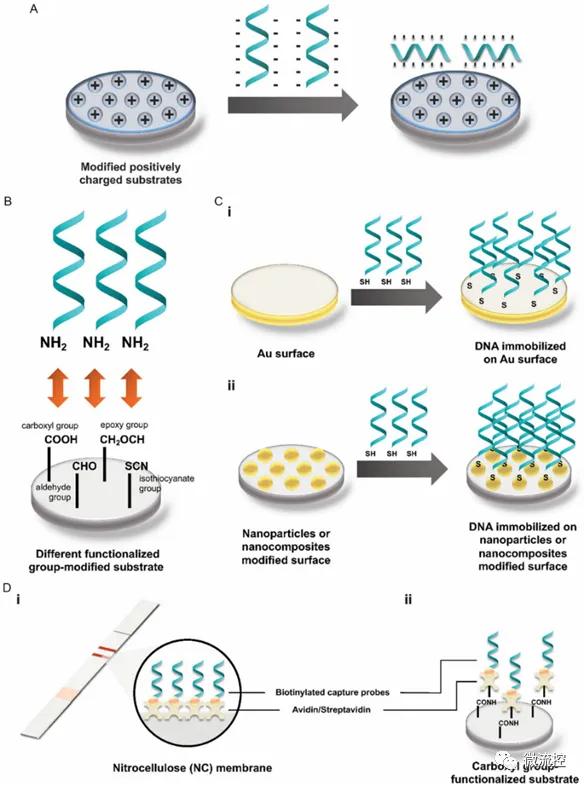
A Schematic representation of the DNA probe fixation procedure
The continuous advances in DNA microfluidic chip technology hold great promise for the development of the next generation of POCT, and the advantages of integrated sample preparation processes and rapid result generation can further improve the therapeutic effect of patients and contribute to the effective management of the disease.
However, while DNA microfluidic chip technology, including point-of-time diagnostics, has made significant progress, it is still a long way from commercialization.
First, as DNA microfluidic chips move from the laboratory to practical application scenarios, their reliability becomes even more important. At the same time, the large-scale, cost-effective manufacturing of DNA microfluidic chips is also a major obstacle. In addition, there are still many challenges in realizing the clinical transformation of DNA microfluidic chips with application potential, including device standardization, effectiveness analysis and validation, ethical related issues and subject selection in clinical trials. Therefore, collaboration between researchers, clinicians, and businesses is crucial.
Overcoming the above challenges is a prerequisite for DNA sensors to reach their full potential and have a significant impact on applications such as healthcare and environmental monitoring. Advances in miniaturization, automation, and analytical integration technologies promise to address the above challenges and streamline relevant clinical workflows.
Paper link: https: / / doi.org/10.1002/anbr.202300058
Our product recommendation:
1.360056-45-7 https://www.bicbiotech.com/product_detail.php?id=5490
2.1190314-06-7 https://www.bicbiotech.com/product_detail.php?id=5491
3.345642-87-7 https://www.bicbiotech.com/product_detail.php?id=5492
4.847490-69-1 https://www.bicbiotech.com/product_detail.php?id=5493
5.75677-02-0 https://www.bicbiotech.com/product_detail.php?id=5494




