Written by Wang Cong
Editor: Wang Duoyu
Scheduled by water
Differentiation of human pluripotent stem cells could provide new therapeutic options for a range of hematopoietic disorders. For example, hematopoietic stem cells (HSC) from patient-derived induced pluripotent stem cells (iPSC) can avoid donor-host mismatch causing graft-versus-host disease (GvHD), and GvHD is the leading cause of death in patients undergoing mismatched allogeneic transplantation. HSC generated from gene-edited iPSC can be treated by correcting the genetic etiology of blood disease (e. g., bone marrow failure syndrome). The use of gene-edited iPSC-derived cells simulating hematopoietic development or disease can accurately recapitulate abnormal hematopoiesis, thereby facilitating the development of more effective therapies. However, regenerating HSCs from pluripotent stem cells is challenging.
On 2 September 2024, researchers from the Royal Australian Children's Hospital and the University of Melbourne published a research paper entitled: Long-term engrafting multilineage hematopoietic cells differentiated from human induced pluripotent stem cells in the Nature subjournal Nature Biotechnology.
The research made a new breakthrough in the world's first, overcome the main obstacle of human blood stem cells, success in the laboratory with the human blood stem cells —— iPS cell source of hematopoietic stem cells (iHSC), these stem cells can produce very similar to human embryo red blood cells, white blood cells and platelets, for the laboratory culture cells for blood stem cells and bone marrow transplantation paved the way. This study may soon bring personalized treatment options for children with leukemia and bone marrow failure.

The first author, Elizabeth S. Ng says, based on this study, we can extract any cells from patients, reprogram them into stem cells, and then convert them into matched blood cells for transplantation, which will have a huge impact on the lives of these vulnerable patients.
Previously, human blood stem cells that can be transplanted into animal models of bone marrow failure have not been cultured in the laboratory to produce healthy blood cells. The new study has developed a workflow that produces transplanted blood stem cells very similar to those found in human embryos. More importantly, these human cells can be produced at the scale and purity required for clinical application.
Here, we established an experimental protocol for induced pluripotent stem cells (iPSC) differentiation, generating CD34 + hematopoietic implantation (MLE) hematopoietic stem cells, which the team called iPS cell-derived hematopoietic stem cells (iHSC). The key elements of the protocol include the defined medium and cryopreservation of CD34 + cells to accommodate future clinical applications.
The results reveal that the MLE capacity of CD34 + hematopoietic cells in immunodeficient mice depends on the timing of Wnt ligands, retinoid precursors, and vascular endothelial growth factor (VE GF), reflecting the role of these molecules in the specialization of the hematopoietic system. This study lays the foundation for further analyzing the process of iPSC and finally realizing clinical application.
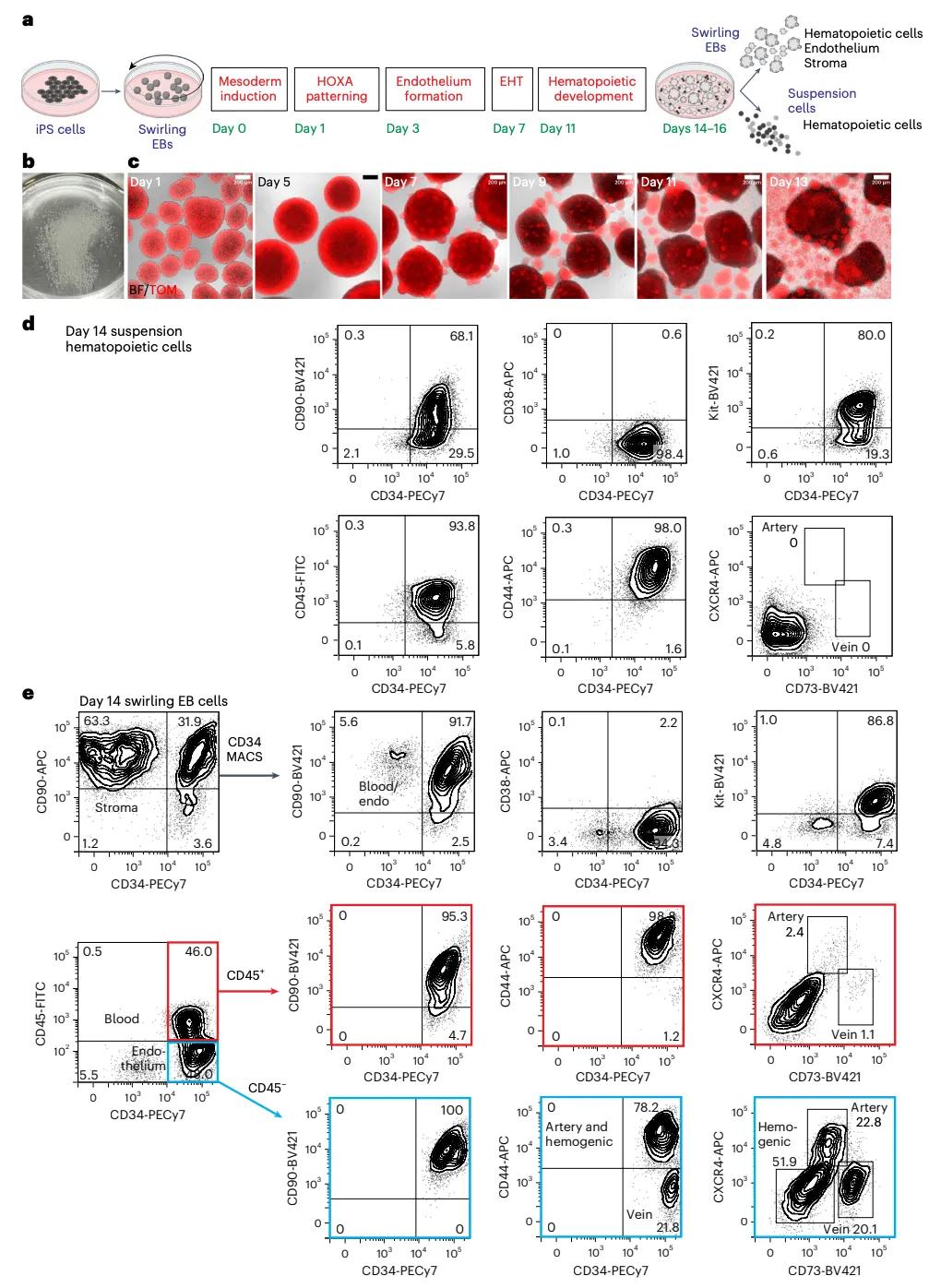
In vitro hematopoietic differentiation of iPS cells
The team transplanted 20 million frozen CD34 + cells differentiated from four independent iPS cell lines into immunodeficient recipient mice to produce multilineage hematopoietic bone marrow engraftment in 25% to 50% of the recipient mice. These functionally defined, multipotent CD34 + hematopoietic cells, named iPS cell-derived hematopoietic stem cells (iHSC), are functionally similar to the bone marrow produced by cord blood HSCT, which is an accepted criterion for success. This study represents an important step towards the goal of producing hematopoietic stem cells for clinical translation.
The study also found that these laboratory-cultured human HSCs can be successfully transplanted into mice after cryopreservation, which mimics the preservation process before transplanting donor-derived human HSCs into the patient.
The team says red blood cells are essential for oxygen transport, and white blood cells are our immune defense system, and platelets stop us from bleeding by causing coagulation. Understanding the development and function of these blood cells is like deciphering a complex puzzle. By refining the stem cell approach that simulates normal blood stem cell development in our body, we can understand and develop personalized treatments for a range of blood diseases, including leukemia and bone marrow failure, thus bringing new therapeutic options for these serious blood diseases.
Professor Andrew Elefanty, the corresponding author of the paper, said that while blood stem cell transplantation is usually the key to saving children from blood diseases, not all sick children can find an ideal matched donor. Immune cells from mismatched donors can attack the patient's body tissue during the transplant, leading to severe disease and even death. The development of personalized, patient-specific blood stem cells can avoid these complications, address the donor shortage problem, while combining with gene editing, helping to fundamentally correct the pathogenic causes of blood diseases.
Finally, Professor Andrew Elefanty said the next phase of work is likely to begin in about five years, with a phase 1 clinical trial to test the safety of using these laboratory-grown blood cells in humans.
Paper link:
https://www.nature.com/articles/s41587-024-02360-7
The company's product recommendation:
1.39684-80-5 https://www.bicbiotech.com/product_detail.php?id=5495
2.51719-65-4 https://www.bicbiotech.com/product_detail.php?id=5496
3.506-68-3 https://www.bicbiotech.com/product_detail.php?id=5497
4.59214-70-9 https://www.bicbiotech.com/product_detail.php?id=5498
5.137076-22-3 https://www.bicbiotech.com/product_detail.php?id=1330




