In November, nitroquintinib was successfully launched in the United States, at a price more than 20 times that in China, confirming the strong payment ability of the American pharmaceutical market. On the other hand, since the first approval of Zebutinib in the United States in 2021, only 6 domestic innovative drugs have been successfully approved by the FDA to land in the US market.
For most drug companies, landing in the US market is a challenging target, so they are focusing on the secondary market. Indian drug companies, for example, began to enter the Middle East and North Africa (MENA) market several years ago. In particular, the emergence of the epidemic has made the regional government pay more attention to the development of the pharmaceutical industry and seek autonomy and control.
It is worth noting that Saudi Arabia, the United Arab Emirates, Qatar, Kuwait, Oman and Bahrain have formed the Gulf State Cooperation Council (GCC), which have formulated a number of policies to encourage the transfer, investment and localized manufacturing of biomedical technologies. In the stage of Chinas innovative drugs towards internationalization, although the European and American markets are the competitive highland, the countries in the Middle East and North Africa also have a large number of unmet medical needs. These regions will also become another road for Chinese pharmaceutical companies to go overseas.
Money and lack of medicine is the norm
Mismatch with the economic strength is the status quo that the medical demand in the MENA region is far from being met.
Last years World Cup in Qatar gave the world an intuitive view of the wealth of the Middle East. The $220 billion World Cup was five times the past seven tournaments, while Qatars GDP was only seventh in the Middle East. As the Middle East gradually moves to the center of the world stage, its high GDP level and huge customer group make the Middle East market more and more valued by multinational enterprises.
According to the data of the World Bank, at present, the MENA region has nearly 500 million people and maintains a long-term growth trend. The per capita GDP is at the level of middle-income countries, among which the total per capita income of the "six Gulf countries" has reached the worlds leading level. In recent years, with the growing population in the Middle East, the local healthcare market is also increasing under the influence of factors such as increasing expenditure on medical services and increasing burden of chronic diseases.
For example, the International Diabetes Federation (IDF) has reported that the prevalence of diabetes in the MENA region has reached 16.2%, and the expected increase of the number of diabetes patients ranks the second in the world, and is expected to reach 136 million in 2045. In addition, obesity is also a serious problem in the region, with MENA accounting for 18 of the top 50 countries with high obesity rates. By 2030, deaths from non-communicable diseases such as diabetes will account for 87 percent of all deaths in the Gulf states and 81 percent outside the Gulf states, according to the World Bank.
Due to the large economic differences in different countries in MENA region, the demand for drugs also varies greatly. For example, due to the high per capita GDP brought by rich oil resources, Saudi Arabia has a high overall consumption level and stronger payment ability, preferring original drugs and patented drugs, the demand for generic drugs is not high, and some economically affluent groups are more inclined to go to Europe and the United States for medical treatment.
On the contrary, on the other hand, Egypt in North Africa has a mainstream market of generic drugs. Although there are many domestic pharmaceutical companies including Egypt International Pharmaceutical Industry (EIPICO), Southern Egypt Pharmaceutical Industry (SEDICO) and Medical United Pharmaceutical, the Egyptian pharmaceutical industry is heavily dependent on the import of API because it has not developed a perfect chemical system and supply chain.
The International Pharmaceutical Raw Materials and Pharmaceutical Industry Exhibition held in Egypt in September focused on pharmaceutical raw materials, pharmaceutical machinery, packaging materials, laboratory equipment and other products. A total of 208 enterprises participated in the exhibition, including nearly 80 Chinese enterprises, accounting for nearly 40% of the total number of exhibitors. As the worlds major producer of API, Egypt is also an important export market for China. In September this year, Northeast China Pharmaceutical reached a cooperation with Muscat ChangmingInvestments to invest and build a factory in Oman to realize the localized production and sales of its products in the MENA market.
According to IQVIA data, the global pharmaceutical market is expected to grow at a compound annual growth rate of 3%~6% to about $1.9 trillion by 2027, among which MENA region is expected to grow by 35%~55% in the next five years, which is expected to become an emerging blue ocean pharmaceutical market.
In response to surging demand for drugs, the GCC has introduced a series of policies aimed at boosting local pharmaceutical capabilities. The export of domestic API, pharmaceutical machinery and laboratory equipment is just the beginning. With the expansion of this blue ocean market, pharmaceutical companies from all countries are involved.
Introduce the industry and upgrade the local pharmaceutical capacity
In addition to the energy industry, the MENA region is vigorously developing the pharmaceutical industry.
MENA region multiple countries developed including rapid examination and approval, registration and the property drug price preferential measures to encourage the pharmaceutical industry localization, and the multinational drug companies cooperate with local manufacturers to establish a joint venture to provide a lot of incentives, prompting multinational drug innovation can quickly and widely into the local market.
For example, Pfizer set up a production base in King Abdullah Economic City (KAEC), one of the four major special economic zones in Saudi Arabia. As a recognition of this investment, Pfizer obtained a trade and investment license from the Saudi General Investment Administration (SAGIA). This gives Pfizer 100% ownership of the Saudi business, including the import and export of products and various trade businesses, to provide quality innovative drugs and essential medicines directly to the Saudi market.
Pfizer is committed to launching about 16 drugs at its production site to meet Saudi Arabias needs in five therapeutic areas: cardiovascular, pain, anti-infective, urology and neurology. Not only production, with the development of the enterprise, Pfizer will also transfer professional knowledge and technology to the local market, and cultivate more professionals while creating jobs.

In recent years, some multinational pharmaceutical companies have cooperated in MENA region, with the data derived from IQVIA and public information
AstraZeneca has signed a cooperation agreement with Saudi Pharmaceutical Industry and Medical Devices Corporation (SPIMACO) to serve the localized production of AstraZeneca pharmaceutical products. Astrazeneca will commission SPIMACO to produce local high-demand drugs in the cardiovascular, diabetic and gastrointestinal sectors, and astrazeneca will quickly reach its products to millions of local patients. SPIMACO We have also improved our ability from this cooperation and prepared ourselves for the development of the national market.
Not only Saudi Arabia, but also other MENA countries are also actively introducing multinational pharmaceutical companies.
As one of the most developed markets in the Middle East, the UAE has a large medical demand, and its per capita medical spending is also in the front row. Before that, more than 70% of the UAEs drugs were imported from Europe and the US. Therefore, the UAE has a strong desire to develop the pharmaceutical industry and bring it in line with the international pharmaceutical production standards.
For example, GlaxoSmithKline collaborated with UAE-based pharmaceutical company Neopharma, and Neopharma became the third-party manufacturer responsible for handling the secondary packaging process (final manufacturing phase) of six prescription drugs in the UAE. In addition, Neopharma has reached a partnership agreement with Merck to localize Mercks diabetes drugs.
With government support, multinational drug companies have moved more frequent in the Middle East over the past two years.
In July 2023, Sanofi partnered with local Saudi biopharmaceutical companies Lifera and Arabio to promote local vaccine production and share expertise in making seven key vaccines. This is in line with Saudi Arabias goal of wanting to strengthen its biopharmaceutical capacity through technology transfer and workforce development.
In June, in Abu Dhabi, the Ministry of Health launched a partnership with Lilly and EbVie, laying the foundation for clinical research, real world evidence, healthcare technology, personalized medicine and genomics. In addition, British pharmaceutical giant AstraZeneca and diagnostics company Virax Biolabs also moved to the Dubai Science Park this year.
In 2022, the Saudi Ministry of Investment reached an agreement with Novartis to work on cell and gene therapy, technology transfer and investment in clinical research, to improve the Arabias biopharmaceutical capacity and reduce healthcare budget pressures. In the same year, the Saudi Investment Ministry also reached an agreement with British pharmaceutical company GlaxoSmithKline to further strengthen Saudi capabilities in the healthcare and life sciences sectors.
Overall, for a certain economic strength of countries in the Middle East, and big pharmaceutical companies cooperation, commissioned production is just the beginning, local pharmaceutical meet local demand is only short-term goals, they prefer to through knowledge technology transfer, let drug production follow global production technical standards and efficient control method, thus have the technical ability of producing complex drugs, to lay a good foundation for the future industrial upgrading, and invest biotechnology as a pillar industry in the future.
But the road of industrial upgrading is doomed to be difficult, and this also gives Chinese enterprises the opportunity.
The road to industrial upgrading needs more participants
In the process of industrial upgrading, many areas have substantial room for growth, waiting for the participation of Chinese enterprises.
With the launch of the cooperation, the MENA pharmaceutical market has maintained rapid growth, including insulin glargine, infliximab, recombinant human propin, filgrastim and other drugs occupying the top of the sales charts. However, the cooperation is not smooth. Taking Saudi Arabia as an example, the communication with multinational pharmaceutical companies for vaccine, blood products and mAb technology introduction and localization has not been smooth in the past few years.
Pharmaceutical companies are more willing to export in finished products for commercial interests, and have limited support for biotechnology transfer, local production and industrial upgrading of local pharmaceutical industry. Therefore, Saudi Arabia and other countries are starting to find more partners, such as pharmaceutical companies in India and South Korea. Compared with them, Chinese pharmaceutical companies are not inferior in the innovation ability and product pipeline richness of biological drugs, so they have enough ability to "go global".
There are plenty of untapped opportunities in the MENA market, with the Gulf Cooperation Commission (GCC) alone having more than 160 health care projects with a total value of more than $50 billion. In the future, there will be several major areas with high growth space.
The first is genomics. In response to the rising incidence of genetic diseases, Saudi Arabia has launched the Saudi Human Genome Project (SHGP) to accelerate the diagnosis of genetic diseases, expand the genetic databases and enhance the understanding of genetic diseases. Qatar has set up the Qatar Genome Project (QGP) to study genomics and genetics in the Middle East, with a focus on precision medicine and research capacity building. The UAE also launched the National Genome Strategy in March, aiming to establish a legal framework to support genome initiatives to improve public health and personalized medicine needs.
As a result, the demand for sequencing technology in the region continues to increase, with some companies responding to the growing demand by launching new technologies and expanding their operations in the Middle East. In April, genetic technology company Malaysian Genomics moved into the Middle East. In June, CENTOGENE formed a joint venture with Lifera, a biopharmaceutical company arm of the Saudi Public Investment Fund (PIF), to enhance the use of leading data-driven multi-omics testing. The Illumina, meanwhile, has launched operations in Dubai.
In September this year, Genalive, an independent clinical laboratory jointly established by BGIs Saudi subsidiary and Tibbiyah Holding, a wholly owned subsidiary of Saudi Al Faisaliah Group, officially opened. At the opening ceremony, Yin Ye, CEO of BGI Group, said: " By combining the advantages of BGI and Tibbiyah, we can develop more potential of genetic testing so that Saudi healthcare workers have the tools needed to provide personalized care for patients. In the future, BGI will bring more Chinese technologies to the Middle East market through Genalive.”
In addition, berry gene also through subsidiary and Saudi agilan brothers medical company (Ajlan & Bros Medical Company) signed a cooperation agreement, a joint venture in Saudi Arabia integration technology, scientific research, industry and strategic resources, the noninvasive prenatal testing (NIPT) and other genetic testing products into Saudi Arabia and the Middle East market, provide accurate genetic disease diagnosis and treatment plan, in the future, the two sides will also start including in the local genetic testing laboratory.
Then there is precision medicine, which Abu Dhabi last year launched the regions first tumor personalized precision medicine program, using genomics to change diagnostics, drug treatments and prevention methods. As early as 2020, Dubai opened an advanced Genomics center at the Al Jalila Childrens Hospital, providing genetic testing and counseling. Dubai will also host the first Middle East Precision Medicine Exhibition and Summit in 2024, demonstrating the regions determination to promote precision medicine in the healthcare sector.
For Chinese enterprises, cell and gene therapy (CGT), as a new generation of precision medicine, has a small generation gap at home and abroad, and even some underlying technologies are still in the leading state. Some companies with ideal pipeline progress and positive clinical data, such as making valuable products, are highly attractive to the MENA market, which is in high demand in oncology, rare diseases, autoimmune diseases and other fields.
Finally, anti-aging research, as part of Saudi Arabias Vision 2030, is to increase the average life expectancy from 74 to 80 years. To that end, Saudi Arabia has established the Hevolution Foundation (composed of the words health and evolution) to fund universities and start-ups in the development of anti-aging drugs. Saudi Arabia has created an annual budget of over $1 billion for the Hevolution Foundation. In September, Abu Dhabis Mubadala Fund participated in the financing for Swiss biotech company Rejuveron Life Sciences AG, a company focused on research and development to prevent and cure diseases that cause aging. The company will open an office in Abu Dhabi and has begun working with local universities and hospitals to promote research on ageing in the Middle East.
Chinese enterprises with technology accumulation, a certain scale in China and a willingness to internationalize are more likely to stand in the Middle East. They enter the local market through technology localization or technology transfer. It is also a growth path to implement business, cultivate local talents and develop local financing development.
Look around the world and pursue cutting-edge technology
However, the introduction is not enough. Middle East capital has launched the "banknote ability" and taken the initiative to go out and join the global frontier wave of biotechnology.
Take Qatar as an example. Its Qatar Investment Authority (Qatar Investment Authority, QIA) was founded in 2005 with assets under management of about $500 billion and is one of the worlds top 10 sovereign wealth funds. Its investment style has also developed from the past energy industry to diversified investment, and biotechnology and life science are its important investment areas. QIA alone participated in several Biotech financing.
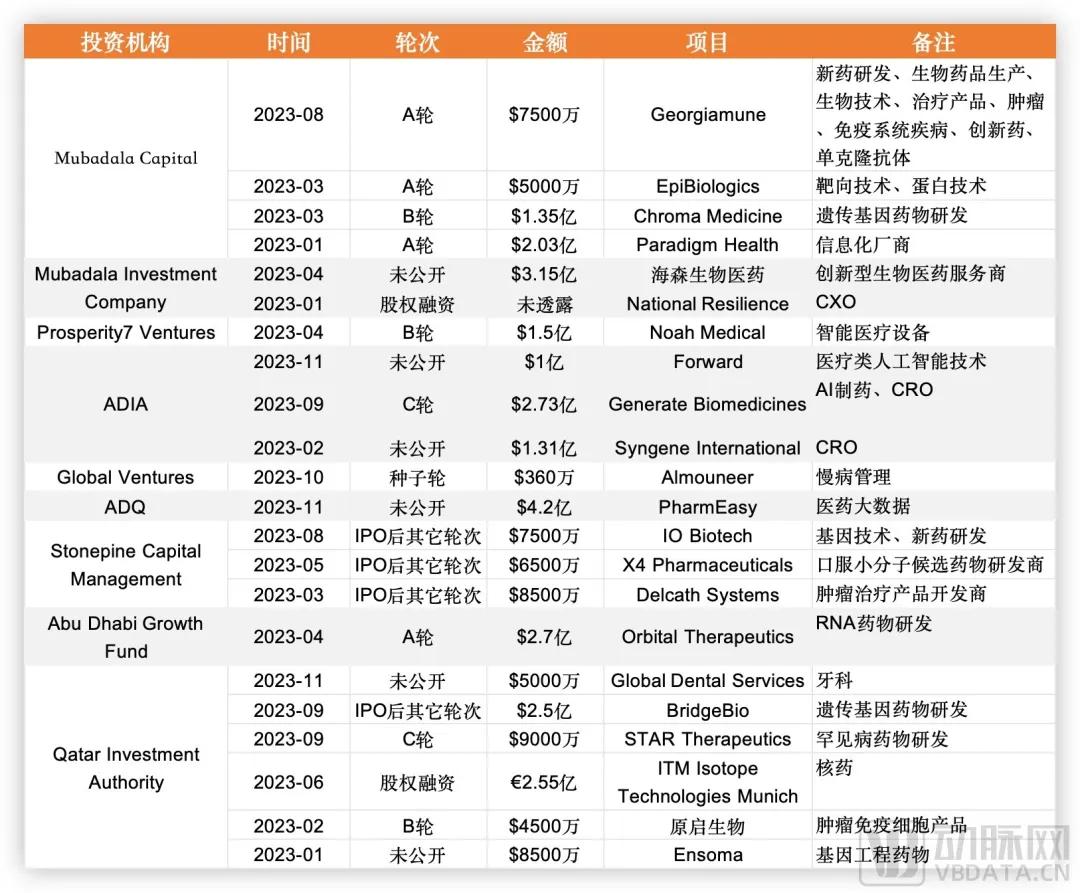
This year, some Middle East capital has participated in financing projects, and the data is derived from artery orange
Like Qatar, many Middle Eastern countries, although rich in oil and gas resources, are no longer satisfied with the structure of their own resource-based economy, and have been trying to transform themselves into a technology-based and sustainable economic structure. Biotechnology and new energy industries are the future they are betting on.
From the investment style of Middle East Capital in the past few years, in addition to the pursuit of innovative drugs, but also to see the development of new technology and the introduction of new technology to upgrade the local medical industry. For example, in the digital transformation of health institutions, according to the plan, GCC countries will upgrade about 30% of domestic medical institutions in the next decade. Among them, Saudi Arabia and the United Arab Emirates will become the leaders in this wave of digital transformation due to their good consumer base.
On December 9, the Saudi Ministry of Investment held an investment conference in Guangzhou, hoping to strengthen the cooperation between Guangdong enterprises and Saudi Arabia, promote the innovation and talent training of Saudi smart city, artificial intelligence and big data, and promote the development of digital transformation. At the same time, Guangdong will actively support the partnership of Saudi PIF and other sovereign wealth funds with relevant funds in Guangdong and increase investment in emerging industries.
In addition, the application of artificial intelligence is also the key development direction of Middle East Capital, including image-based artificial intelligence and AI pharmaceuticals. In this respect, domestic enterprises also have enterprise to participate. In early July, Jingtai Technology, Nuohui Health and many other companies signed a memorandum of cooperation to enter the Saudi market and join the China-Saudi Arabia Enterprise Federation. In order to layout the Middle East market, Jingtai Technology has visited the region for many times, and finally chose Saudi Arabia, which has a strong demand for AI research, as the first stop to go out to the Middle East. In the future, Jingtai Technology will not only use artificial intelligence in medical and health tracks, but also hope to make breakthroughs in new tracks such as materials science.
In February, British Silicon Intelligence (Insilico Medicine), another AI-driven drug discovery company based in the Hong Kong Special Administrative Region, also announced the opening of a generative artificial intelligence and quantum computing research and development center in ABU Dhabi. The company has used AI to develop an experimental drug targeting idiopathic pulmonary fibrosis disease. Medium-term trials are currently underway in the United States and China, with preliminary results expected in early 2025.
During the outbreak, Sinopharm, Consino and Yisheng companies carried out clinical trials of vaccines in Saudi Arabia, the United Arab Emirates and other places, laying a foundation for the continuous cooperation between the two sides. After that, many domestic innovative pharmaceutical companies began to pay attention to this blue ocean market. For example, the PD-1 drug Triplizumab signed an exclusive license and commercialization agreement with Hikma MENA FZE, and developed and commercialized the exclusive license in 20 countries in Jordan, Saudi Arabia, UAE, Qatar, Morocco, Egypt and other MENA regions.
In addition, East China Pharma has cooperated with Julphar, a Gulf pharmaceutical company in the UAE, to grant Julphar the development, production and commercialization interests of its liraglutide injection in 17 countries in the UAE region, Saudi Arabia and other MENA regions. Paikche China has cooperated with NewBridge Pharmaceuticals to promote zebritinib in Kuwait, Bahrain, Qatar, Saudi Arabia, the United Arab Emirates and other countries.
In September this year, Fuhong Hanlin reached a cooperation agreement with PT Kalbe Farma Tbk, granting its exclusive development and commercialization interests in 12 MENA countries, including Saudi Arabia, UAE, Egypt, Qatar, Jordan and Morocco, in the PD-1 anti-Srulumab independently developed by Fuhong Hanlin.
For pharmaceutical companies with international experience, licensing cooperation in the MENA market is only the beginning. Joint venture or investment with local pharmaceutical companies, or technology authorization for local production and marketing, and growing into a leading brand in a disease field or subdivision track in the region will be an important step for growing into a global pharmaceutical company. Domestic innovative vaccines, monoantibodies and other drugs have broad development prospects.
In September this year by astrazeneca and jun league capital support of Chinese medical enterprise delegation to open a trip to the Middle East, including consino, green pharmaceutical, ace creature, Changchun high, cinda biological, Ann, hui health, Innomind, kang lian, Etana, bojie medical, medical technology and jun league capital, covers the pharmaceutical, biotechnology, vaccines, equipment, diagnosis, medical data and capital, and other fields.
Seize the Middle East, or will become the Chinese pharmaceutical companies to break the sea road.* Reference:
●https://www.fiercepharma. com/manufacturing/uae-merck-serono-neopharma-announce-first-pharmaceutical-production-agreement
● IQVIA White Paper November 2020 "Realizing Biosimilar Potential In the Middle East & Africa"
● IQVIA White Paper "Localization of Pharmaceutical Manufacturing in Middle East and North Africa Region"
* Cover image source: 123 rf
The companys product recommendation:
1.16874-75-2 https://www.bicbiotech.com/product_detail.php?id=5510
2.63510-09-8 https://www.bicbiotech.com/product_detail.php?id=5511
3.1368440-33-8 https://www.bicbiotech.com/product_detail.php?id=5512
4.1150114-75-2 https://www.bicbiotech.com/product_detail.php?id=5513
5.886499-83-8 https://www.bicbiotech.com/product_detail.php?id=5514




