Where is the word "overcrowded" best used right now? The answer is in the pediatric departments of the major hospitals.
Winter has come, entering the season of high incidence of respiratory infectious diseases. In many places, the number of outpatient services and emergency departments in hospitals has increased, and the number of children infected with respiratory diseases has increased.
The pathogens of respiratory infectious diseases mainly include novel coronavirus, influenza virus, mycoplasma pneumoniae, etc. This year, respiratory syncytial virus also joined the ranks of the epidemic, causing widespread attention, and once became the hot search.
Ten billion market rise again
Respiratory syncytial virus (RespiratorySyncytialVirus, RSV) is a virus belonging to the Paramyxviridae with a negative single-stranded RNA genome. The virus was first isolated from the respiratory tract of chimpanzees in 1956. Respiratory syncytial virus because of its association with respiratory infections and its ability to fuse infected cells.
The development history of human RSV vaccine is more than 70 years, and repeated failures, which is enough to prove that the development barrier of RSV vaccine is extremely high. However, the market outlook is very considerable. According to the data, the overall global market size of RSV drugs is expected to grow from $1.8 billion in 2020 to $12.8 billion in 2030, with a compound annual growth rate of 21.4%. Therefore, RSV drug is a competition between the major companies, but also the technical route.
Recently, two exciting news about the RSV vaccine came both at home and abroad.
On December 13, Clover Biology announced that the first subjects have been enrolled in a phase I human clinical trial to evaluate the RSV PreF-trimer subunit vaccine candidate (SCB-1019), based on an innovative protein trimerization (Trimer-Tag) vaccine technology platform.
SCB-1019 is a bivalent recombinant protein RSV vaccine candidate containing two F antigens in stable pre-prefusion conformations (subtype A and B subtype RSV F proteins) using the validated Trimer-Tag vaccine technology platform and patented highly stable PreF mutations.
The current phase I clinical trial initiated in Australia is a randomized, placebo-controlled study to evaluate the safety, reactogenicity and immunogenicity of SCB-1019 at different dosage forms and dose levels in populations of young adults and older adults. Safety and immunogenicity results are expected to be published in the second half of 2024.
SCB-1019 is the first RSV PreF vaccine in China to enter the clinical trial stage, which strengthens the leading position in the RSV field and proves the innovative value of the Trimer-Tag vaccine technology platform.
Abroad, on December 12, AstraZeneca announced that it had signed a definitive acquisition deal with vaccine developer Icosavax to buy Icosavax for a total price of up to $1.1 billion.
Through the acquisition, AstraZeneca will acquire the rights to develop Icosavaxs core project, IVX-A12. IVX-A12 is a VLP combination vaccine candidate for potential "first-in-class" for respiratory syncytial virus (RSV) and human metapneumovirus (hMPV) in the elderly.
Icosavax Positive Phase II clinical data for IVX-A 12 were released on December 11, showing that after 28 days of vaccination, IVX-A12 can cause a potent immune response against RSV and hMPV – with or without adjuvant addition.
In addition to the research vaccine, AstraZeneca will acquire ownership of Icosavaxs proprietary virus-like particle (VLP) vaccine technology, which can display high-density, multivalent antigens in a similar viral structure, according to Icosavaxs website.
Icosavax Said that vaccine candidates developed using the VLP platform can induce a stronger and more durable immune response than a traditional antigen-based vaccine.
Shamrock Biology took the lead in the domestic RSV PreF vaccine track with Trimer-Tag platform, while AstrazZeneca entered the market through the acquisition of vaccine companies. The two companies undoubtedly set off a wave in the RSV vaccine market.
The RSV market pattern, both the new and the old relay
In the blue ocean market, there are four major drugs approved for RSV related indications, namely paliizumab (Palivizumab) in AZ, long-acting monoclonal antibody Beyfortus for Sanofi / AZ, GSKs RSV vaccine Arexvy and Pfizers RSV vaccine Abrysvo.
The indications covered by the four products are different: Arexvy is approved for people over 60 years, Abrysvo for people over 60 years and pregnant women at 32-36 weeks gestation; Beyfortus is approved for lower respiratory tract diseases caused by RSV in all infants (long-term, prevention throughout the epidemic quarter); Paliizumab is approved for prevention of severe lower respiratory tract diseases caused by RSV in children and recommended once a month during the RSV epidemic season.
Paliizumab was approved in June 1998, even as a short lasting RSV preventive drug, with past sales of $1.6 billion.
Table: Global marketed RSV drugs
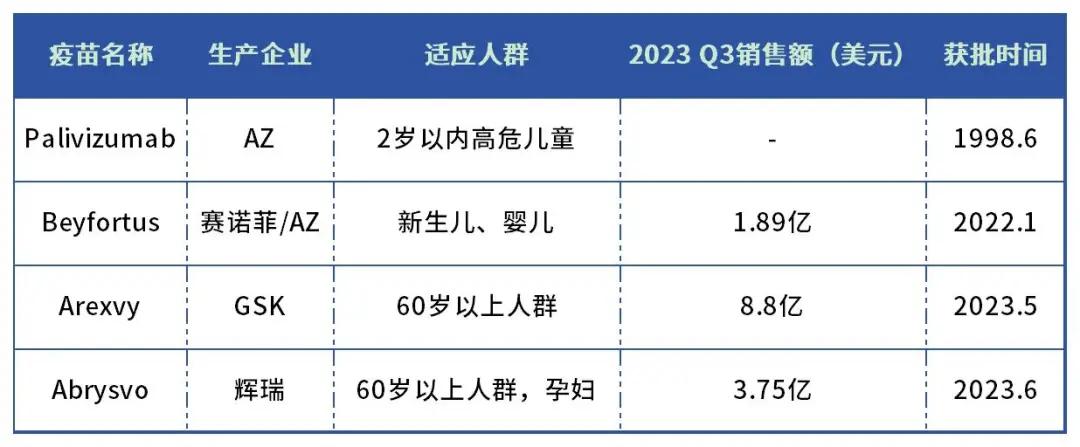
Data source: drug intelligence data, public data collation
Arexvy and Abrysvo are the first year of the RSV vaccine.
Arexvy became the first RSV vaccine to receive FDA approval in May 2023. Abrysvo Approved in June 2023, the second RSV vaccine approved by the FDA, slightly later than Arexvy.
The third quarter of 2023 was the first time that the two products went head to the market. According to GSKs latest third-quarter data, Arexvy accounted for two-thirds of U. S. retail vaccine coverage, selling 709 million euros, or about $880 million. The Abrysvo was Pfizers fastest growing product, selling $375 million in the first quarter, well beating market expectations. Pfizer is extending Abrysvo to the pediatric patient population, while GSK is expanding the target population to the elderly population.
The two RSV vaccines had a combined sales of $1.22 billion in their first quarter, creating the potential to become a $10 billion super blockbuster product.
Compared to GSK and Pfizers two RSV vaccines, Beyfortus, jointly produced by Sanofi and AstraZeneca, focuses on infants, while the former targets people aged 60 and older. That is, there are currently no direct competitor for Beyfortus in the field of infant RSV prevention.
Beyfortus It was approved in the European Union and the UK in October and November 2022, and in Canada and the United States in April and July this year. China, Japan, and several other countries are currently reviewing regulatory applications.
However, shortly after the US market was approved, the Beyfortus was in short supply.
According to the CDC, about 58,000 to 80,000 children under the age of five are hospitalized in the United States each year, and as many as 300 people die from RSV, indicating a huge gap in demand for Beyfortus in the United States.
On October 23, the CDC reminded healthcare providers of the Beyfortus supply shortage and recommended priority use for infants at the highest risk of RSV complications, including infants at 6 months of age or less, and those with heart and lung disease.

Photo credit: CDC CDC website
In response to this alert, pediatricians informed parents that the drug was be available if their infant did not meet these high-risk criteria.
A Sanofi spokesman said that although the demand for Beyfortus has been higher than expected.
To address the shortage, an AstraZeneca spokesman said it was maximizing productivity efficiency and had recently increased production and packaging capacity by adding other production sites.
Beyfortus As a RSV prevention method that can be widely used in newborns and infants, it has become a bright star in the field of RSV prevention. At the moment it is listed, it is similar to the situation of the current two star weight loss drugs, migrutide and tierbase peptide, that is, it is out of stock on the market.
But both Novo Nordisk and Eli Lilly are rapidly expanding their production capacity, constantly investing in factories or increasing their outsourcing efforts.
So Sanofi and AstraZeneca may refer to Novo Nordisk and Eli Lilly.
epilogue
RSV infection disease burden is heavy, and the demand gap of RSV vaccine is huge. So far, with the exception of the RSV Pre-F candidate, no RSV PreF vaccine has entered clinical trials, and there are opportunities for differentiated competition worldwide.
Seasonal attributes and large market demand, RSV vaccine is expected to become the blue ocean field of vaccine segmentation in the future.
reference material:
1, clover biological official micro
2, extremely short supply, vaccine one brother win hemp; gazelle society
Our companys product recommendation:
1.84758-81-6 https://www.bicbiotech.com/product_detail.php?id=5515
2.53956-05-1 https://www.bicbiotech.com/product_detail.php?id=5516
3.13404-22-3 https://www.bicbiotech.com/product_detail.php?id=5517
4.13288-57-8 https://www.bicbiotech.com/product_detail.php?id=5518
5.5978-22-3 https://www.bicbiotech.com/product_detail.php?id=5519




