Cancer immunotherapy- - -mRNA
The mRNA therapy has become a frontier research field of cancer immunotherapy because of its superior ability to express known proteins in vivo. In particular, through small doses of mRNA delivery, antigen-presenting cells (APC) can synthesize mutant neoantigens and polyantigens and present epitopes to T lymphocytes to trigger antitumor effects. Moreover, expression of receptors such as CAR, TCR, CD134 in specific immune cells as well as cytokines, interferons, and antibodies can enhance the immune response against tumors. However, clinical translation based on mRNA anticancer strategies is limited due to insufficient endosomal escape efficiency of mRNA delivery to target organs or cells and to mRNA.
In recent years, some progress has been made in mRNA-based cancer immunotherapy, which can be roughly divided into the modification of mRNA structures and the development of delivery systems (especially lipid nanoparticle platforms).

Recently, Professor Yu Haijun, Shanghai Institute of Materia Medica, Chinese Academy of Sciences, and Professor Xu Zhiai, East China Normal University published a review article entitled "From structural design to delivery: mRNA therapeutics for cancer immunotherapy" in Exploration, highlighting strategies to overcome the bottleneck of mRNA-based cancer immunotherapy, such as mRNA instability, innate immunogenicity and lower expression efficiency. In addition, also emphasized the design of the optimal delivery platform to the mRNA transport to specific sites, classified by target organs and cells (figure 1), discussed the function of specific molecular structure in the delivery system, especially with targeted delivery characteristics, also expounds the current mRNA based cancer immunotherapy clinical application challenges and opportunities.
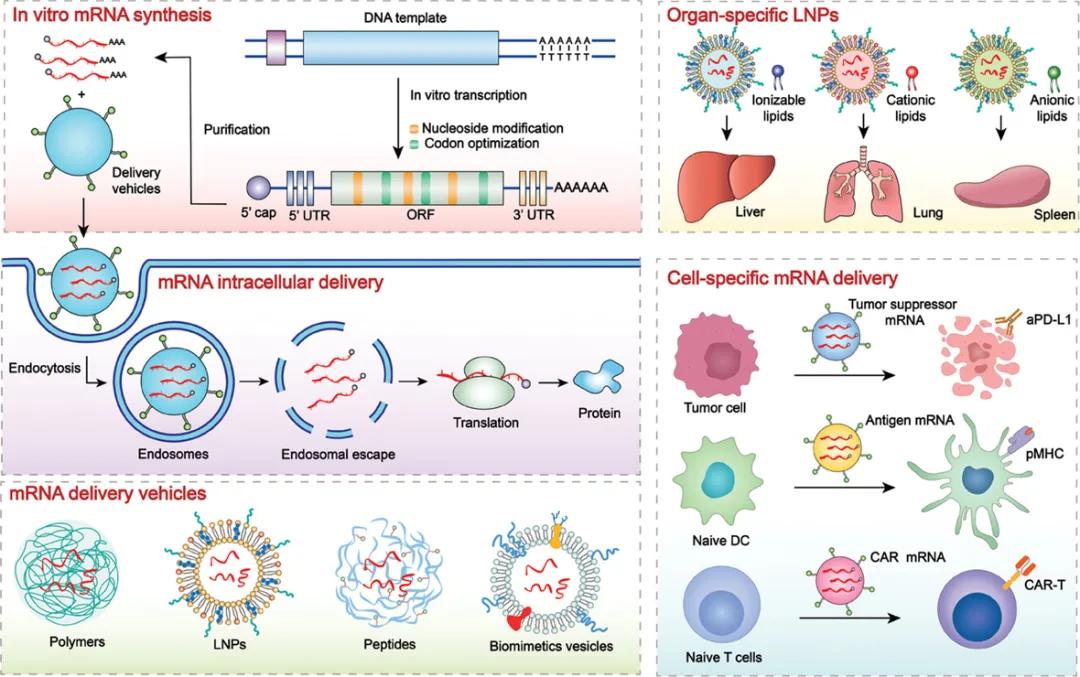
▲ Figure 1 Schematic diagram of the principle of mRNA chemical modification and tissue / cell targeted delivery
Molecular design of mRNA-based cancer immunotherapy
The mRNA is a negatively charged single-stranded RNA containing genetic information transcribed from DNA and relies on the ribosome to translate into proteins for specific life functions. This fragile macromolecular structure determines the mRNA instability in an in vivo environment rich in various peptides and enzymes. In addition, polyanionic mRNA can repel negatively charged cell membranes (CM), making it difficult to enter the cell. The IVT method uses bacteriophage T7 RNA polymerase (T7 RNAP) to synthesize mRNA with high fidelity under the guidance of the linearized DNA template, but due to the gap between the natural source mRNA and the synthesized mRNA, the protein expression of mRNA is not effective enough, and further modification of the mRNA structure and nucleotides is required. Furthermore, the low efficiency of endosomal escape significantly impaired the efficacy of RNA drugs. For example, the FDA-approved DLin-MC3-DMA LNP only mediates the release of 1 – 4% of the RNA into the cytoplasm.
Innate immunogenicity of mRNA is another consideration. Exogenous mRNA acts as an immunogen rather than as a therapeutic agent, which is determined by the innate immunogenicity of the mRNA. APC specifically recognizes IVT mRNA through pattern recognition receptors (PRR) such as Toll-like receptors (TLR), which stimulates the secretion of type I interferons (IFN-I, including IFN- α and IFN- β) and proinflammatory factors, thus significantly impairing the process of protein expression of mRNA. Moreover, IFN-I is both beneficial and detrimental for the performance of cancer immunotherapy. It promotes the maturation of dendritic cells (DC), antigen presentation to T cells, and the activation of CD8 + T cells. On the other hand, IFN-I can increase the number of Treg and Th 17 cells, induce the intratumoral infiltration of myeloid-derived suppressor cells (MDSC), and promote the immune escape of tumor cells.
In the initial stage of in vivo mRNA therapy, mRNA injected into the body causes a series of heterologous immune responses and is lysis by the immune system, just as the body is invading the virus, which greatly limits the application of mRNA therapy. Therefore, technological breakthroughs to overcome mRNA immunogenicity are crucial. In 2005, Katalin et al found that replacing uridine with pseudouridine reduced the risk of mRNA DC activation, not only protected synthetic mRNA from immune elimination, but also significantly increased the efficiency of protein expression. This finding addresses concerns about mRNA-based therapies, thus opening the clinical application of mRNA. And won the Nobel Prize in Physiology this year. In addition to nucleoside substitutions, IVT-mRNA structural modifications include 5 ′ Cap, poly (A) tail and UTR.

▲ Figure 2 schematic of mRNA structural modification, adjuvant addition, nucleoside modification and codon optimization for mRNA-based cancer immunotherapy
Another strategy to improve mRNA stability and protein expression efficiency is to optimize the UTR sequence, and the UTR of mRNA can be divided into 5 ′ and 3 ′ ends. The 5 ′ UTR is close to the start codon of the mRNA and can affect the rate of ORF translation. A report suggests that a shorter 5 ′ UTR without complex secondary structure and start codons (e. g. AUG and CUG) facilitates the initiation of mRNA translation processes, and these points merit consideration in designing mRNA vaccines. The 3 ′ UTR is a common regulatory element that also affects the mRNA expression efficiency. Some investigators have screened and obtained multiple novel 3 ′ UTR, which significantly increased the protein translation level of mRNA compared with the general 3 ′ UTR.
In addition, by analyzing endogenous gene expression and innovative design UTR sequence, people screened the best combination of 5 ′ and 3 ′ UTR, whose protein expression efficiency is 5 to 10 times higher than endogenous UTR. Furthermore, machine learning can be used to facilitate UTR sequence design. Castillo-Hair et al. developed a convolutional neural network model trained on experimental data, called Optimus 5-Prime, which can be combined with algorithms to design UTR sequences, improve translation efficiency and accelerate the process of developing novel UTR in mRNA therapy.
In addition, nucleoside modification and codon optimization of mRNA are also important for efficiency improvement, such as 5-methyl cytidine (m5C), N6-methyl adenosine (m6A), pseudouridine (Ψ), 5-methoxyuridine (5 moU) and 1-methyl pseudouridine (m1 Ψ) are the main modified nucleotides used for mRNA remodeling, which not only improve the efficiency of protein translation, but also reduce the innate immune activation of mRNA. However, due to the degeneracy of codons, there are hundreds of millions of codon combinations and secondary structures in the amino acid sequence of the same protein, leading to the screening of mRNA nucleotide sequences with optimal stability and translation efficiency. Recently, Baidu launched an artificial intelligence (AI) and dynamic programming algorithm called LinearDesign, which can shorten the screening process, allowing the discovery of mRNA with the best sequence in about 10 minutes.
A delivery system for cancer immunotherapy with mRNA
The naked mRNA is susceptible to degradation by the ubiquitous RNase in vivo, and the mRNA needs to be encapsulated in a delivery platform to enhance its endosomal escape efficiency, playing a crucial role in the subsequent antigen presentation process. Multifunctional delivery strategies confer mRNA the ability to target specific organs and cells, which can effectively activate APC and stimulate immune-related signaling pathways through adjuvant action, thus significantly improving antitumor efficacy. In general, suitable delivery systems can help overcome the bottleneck of mRNA-based cancer immunotherapy, such as targeted delivery, improved transfection efficiency, and enhanced strength of immune response. The current technology platforms used for mRNA delivery include LNP, hydrogel materials, polymer delivery, and peptide-based delivery methods, as shown in the figure below.
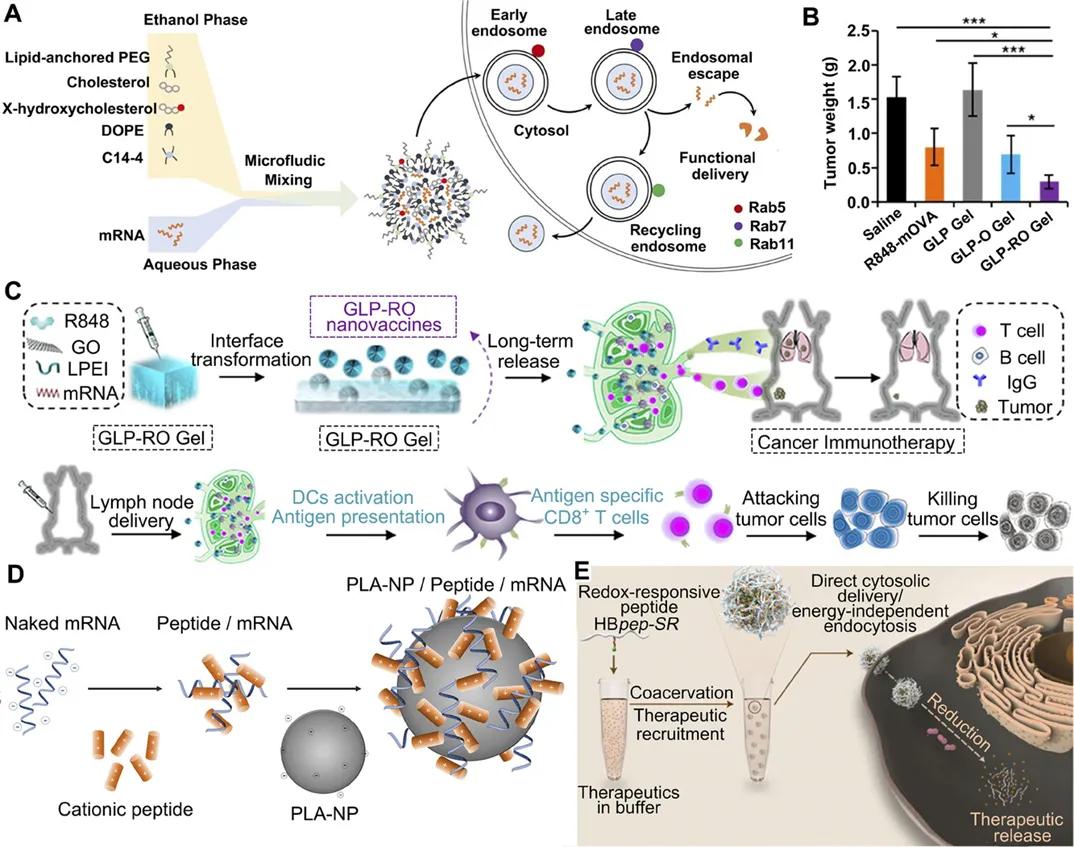
▲ Figure 3 Schematic representation of the LNP, hydrogels, polymer, and peptide-based delivery used for mRNA delivery.
A. Engineered LNP with hydroxycholesterol substitution for delivery of mRNA to T cell cancer immunotherapy. B. Plot of tumor weight changes after treatment with GLP-RO Gel and other groups. C. GLP-RO gels were prepared using polyethylenimine and RO hydrogels for persistent cancer immunotherapy. D. Delivery of mRNA to DC with PLA NP and CPP to induce an effective immune response. E. Phase-separated peptides used for mRNA cytoplasmic delivery, with higher stability and high transfection efficiency.
In conclusion, this review details the precise transport of mRNA to specific organs and cells such as tumor cells, DC, T cells, B cells and macrophages, suggesting that the clinical transformation of mRNA-based cancer immunotherapy will benefit from the deep integration of biochemistry in mRNA design, and the adoption of materials and nanotechnology in targeted mRNA delivery is likely to solve the current challenges in clinical practice. Due to the limited space, there will be no too much introduction here. Interested friends can read the original text in detail to learn about the recent frontier progress and clinical application in the field of mRNA cancer therapy.
Original link:
https://onlinelibrary.wiley.com/doi/full/10.1002/EXP.20210146
The companys product recommendation:
1.1184172-53-9 https://www.bicbiotech.com/product_detail.php?id=5520
2.1214323-32-6 https://www.bicbiotech.com/product_detail.php?id=5521
3.876919-08-3 https://www.bicbiotech.com/product_detail.php?id=5522
4.1147014-97-8 https://www.bicbiotech.com/product_detail.php?id=5523
5.1056264-66-4 https://www.bicbiotech.com/product_detail.php?id=5524




