Figure 1. Cyclopeptide drugs approved in 2023
01 Merck D signed a large contract to develop cyclic peptide drugs
In January 2024, Merck signed a $220 million deal with Unnatural Products, indicating further entry into cyclopeptide drug development.
Unnatural Products, a California-based biotechnology company that specializes in using artificial intelligence and combining chemical expertise to develop complex macrocyclic compound drugs. After signing the partnership deal with Unnatural Products, Merck has started using the other party's technology to develop macrocyclic peptide drug candidates, mainly in the challenging field of oncology. Merck has previously called macrocyclic peptides "blondomes" that have many unique features because of their molecular size between small molecules and biological agents.
Macrocyclic drugs have a long history of development, such as the immunosuppressant cyclosporine (Cyclosporine A) and the antibiotic erythromycin. Systematic development of macrocyclic compounds faces a series of challenges including pharmacokinetics, cell permeability, and oral bioavailability, but still inspires pharmaceutical manufacturers to invest more.
Merck has launched a phase III project on MK-0616. MK-0616, a macrocyclic peptide that binds to PCSK 9 (Figure 2), is currently studying its role in reducing low-density lipoprotein cholesterol. MK-0616 is a macrocyclic PCSK 9 inhibitor that showed biological activity to reduce PCSK 9 and LDL cholesterol in once-daily human clinical trials (<300 mg QD). Currently, PCSK 9 targeted therapies can only be achieved by injection, and the cost-effectiveness of reducing cholesterol with a biologics has not been effectively addressed. Oral PCSK 9 inhibitors are the target in this field, and the development of the MK-0616 macrocyclic peptide is expected to achieve a breakthrough in this field.
MK-0616 related expansion content: Merck PCSK 9 inhibitor started phase III clinical, will be the next "lipid reduction" blockbuster drug?

gure 2. Chemical structure of MK-0616
02 Polypeptide antibiotic breakthrough
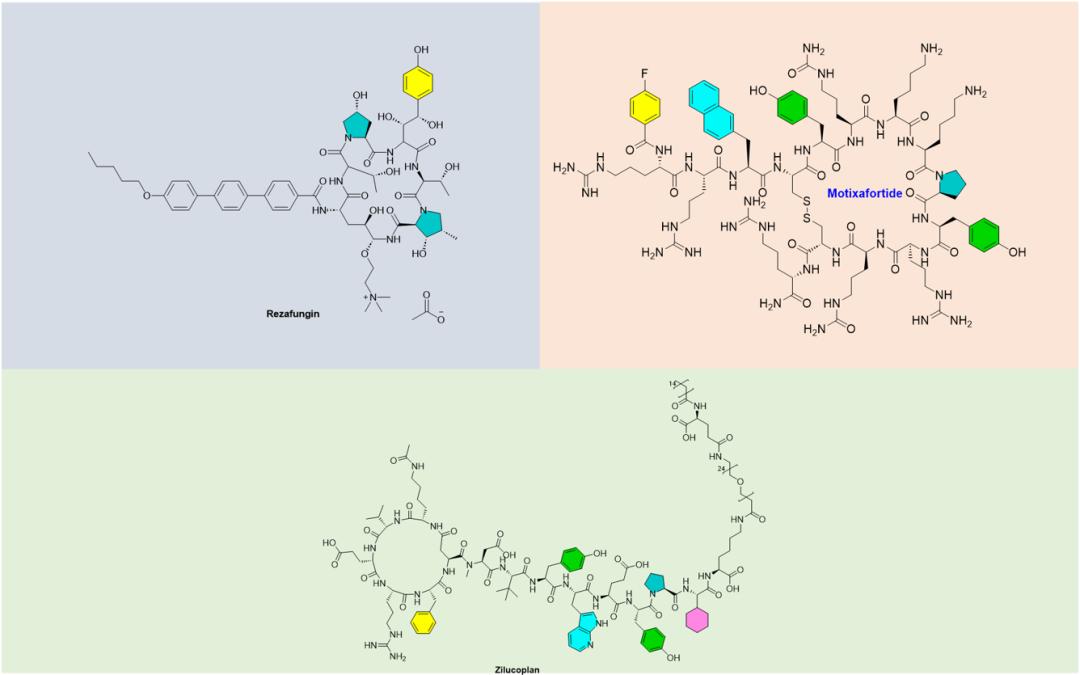
Zosurabalpin (Figure 3) is a cyclic peptide candidate for resistant Acinetobacter pastrilis (Acinetobacter baumannii). Its emergence not only represents the discovery of a completely new type of antibiotic, but also represents a breakthrough in the therapeutic development of Acinetobacter baumannii.
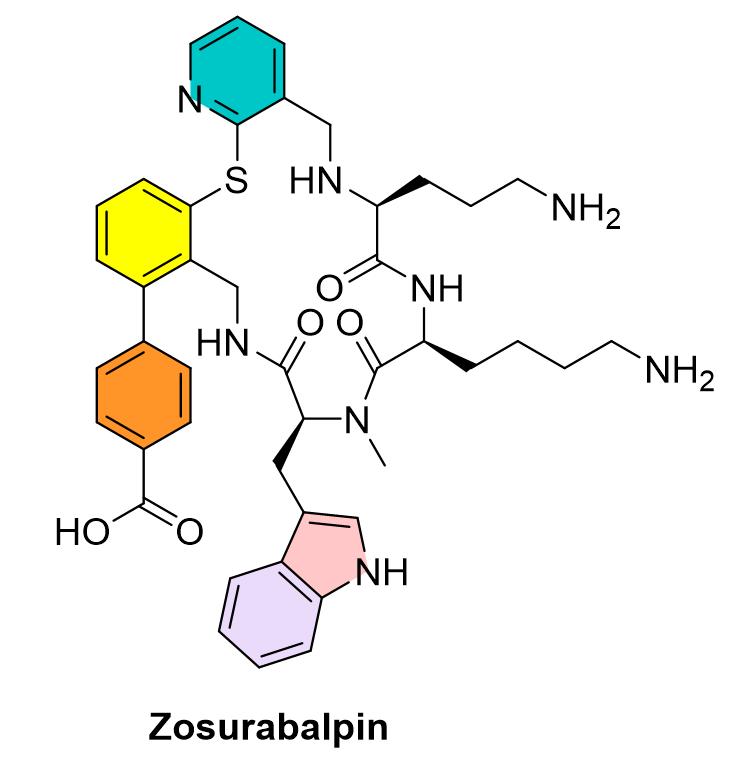
Figure 3. Zosurabalpin Chemical structure
Zosurabalpin Is a macrocyclic peptide antibiotic, acting in a different way from the currently used antibiotics. This novel antibiotic was discovered by a library of 45,000 macrocyclic peptides produced by solid-phase parallel synthesis, directed against a series of human pathogens screens. This new antibiotic has potent activity against Acinetobacter pasteililis (CRAB, Carbapenem-resistant Acinetobacter baumannii), which develops resistance to carbapenem antibiotics (carbapenems). CRAB is a multi-resistant strain that poses serious challenges to treatment because they have developed resistance to multiple antibiotics, including drugs often seen as the "last line of defense" against carbapenems. This makes the treatment of CRAB infection very difficult and sometimes may not be even effectively treated, posing a serious threat to public health. Rized as the first priority pathogen by the World Health Organization, which can cause serious and even fatal infections.
Zosurabalpin Function by preventing the transport of lipopolysaccharide (LPS, lipopolysaccharide). LPS is an important outer membrane component of Gram-negative bacteria. Macrocyclic peptide candidates block the movement of LPS from the inner membrane to the outer membrane by inhibiting protein complexes (transporters) located between the inner membrane and the outer membrane, killing bacteria when LPS accumulates to toxic concentrations (Figure 4).
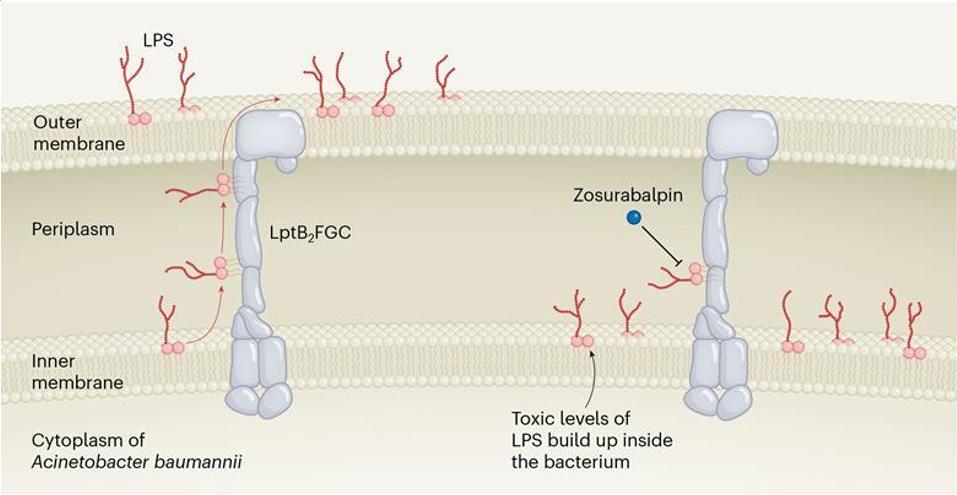
Figure 4. Mechanism of Acinetobacter pahilis, source: Nature
New classes of antibiotics are not frequently present and have been more than 50 years since the new type of antibiotic chemical class with antiacinetobacter pahii activity was approved. Gram-negative bacteria are particularly difficult to kill because their cytoplasmic membrane is surrounded by an outer lipopolysaccharide membrane, preventing the entry of most antibiotics. But the lack of incentives to encourage drug companies to develop new antibiotics also to the resistant bacteria.
As a new type of antibiotic candidate, the macrocyclic peptide zosurabalpin can be used to treat highly resistant CRAB in vitro and in multiple mouse infection models (including sepsis).It has been evaluated in two phase I clinical trials and shows that a single intravenous dose of 10mg to 2000mg zosurabalpin is safe and well tolerated overall, and has attracted the attention of Roche.
03 Oral cyclotides and drug development
Another potential advantage of cyclic peptides is manifested in their pharmacokinetics. Specifically, its unique properties in terms of stability and hydropathy / lipophilic balance, cell penetration, make cyclic peptides a focus in drug development for oral polypeptides. Among the currently available oral peptides, including Cyclosporine A, Voclosporin and desmopressin (Figure 5).
Figure 5. Chemical structures of some marketed oral cyclic peptides
The reason why cyclic peptide has the potential of oral delivery is mainly due to its high biological stability and can resist the degradation of exonuclease. Cyclic peptide in different polar environment can take different spatial structure, help cyclic peptide through the membrane, the cell membrane polar head (polar headgroup) and nonpolar tail (unpolar tail), take different structure and arrangement direction, is advantageous to the cyclic peptide molecules through the digestive tract epithelial cells into the system circulation, relative to the straight chain peptide, its oral bioavailability increased significantly.
In this process, the formation and recombination of hydrogen bonds between the cyclic peptide, between the base oxygen of the backbone amide bond and the hydrogen on the nitrogen, is critically important for the spatial orientation of the cyclic peptide side chain functional groups.
Key parameters for the development of oral polypeptide drugs include molecular weight, number of hydrogen bond donors (HBD), and topological polar surface area (PSA). Most oral peptides have molecular weights less than 700 Da and TPSA less than 200 A2, The number of hydrogen bond donors does not exceed 5. But this is not absolute, for example, Cyclosporine A (CsA; 1203 Da), which has many N-methylated amino acids and can adjust its conformation to limit its polar surface. Internal molecular hydrogen bond interactions of cyclic peptides are critical to reducing TPSA and increasing the oral bioavailability of cyclic peptides.
04 Cyclopeptide drug market size
According to a new report, " Global Constrained Peptide Drugs Market by Peptide Type (Macrocyclic, Stapled), End-User (Hospitals & Clinics, Pharmaceutical Companies) -Forecast 2024-2030 " (Forecast for the global restricted peptide drug market, By peptide type (macrocyclic peptides, Book peptide), End-users (hospitals and clinics, Pharmaceutical Company) -Forecast 2024-2030) Forecast, Restricted peptides (including macrocyclic and book peptides, The latter is also essentially a cyclic peptide) and the drug market will grow to $4.76 billion from $1.22 billion today.




