1. Overview
Irradiation sterilization refers to the method of killing microorganisms using ionizing radiation. Commonly used radiation sources include cobalt-60 (60Co) or cesium-137 (137Cs), γ-rays produced by radioactive decay, electron beams from electron accelerators, and X-rays generated by X-ray devices. This sterilization method directly targets vital macromolecules such as proteins, nucleic acids, and enzymes, causing molecular changes through ionization, excitation, or chemical bond breakage. Indirectly, ionizing radiation interacts with water molecules within microorganisms, inducing water ionization and excitation to produce free radicals that subsequently affect biologically active molecules. The technique boasts high efficiency, strong penetration power, no radioactive contamination or residue, cold sterilization properties, and broad applicability. In the pharmaceutical field, radiation sterilization has gained increasing attention due to its "cold treatment" characteristics, making it an ideal choice for heat-sensitive drugs, excipients, or certain inner packaging materials.
2. Irradiation sterilization related research
After literature review, the information of some products approved for irradiation sterilization at home and abroad is shown in the following table:
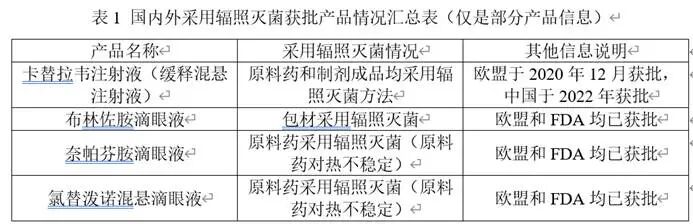
Currently, there are not many drugs approved for irradiation sterilization in China, possibly due to some misunderstandings about irradiation sterilization in the country. When it comes to irradiation sterilization, the first reaction is nuclear radiation and residual radiation. In reality, irradiation sterilization leaves virtually no residue and has become one of the internationally recognized terminal sterilization methods with high sterility assurance levels. At present, regulatory agencies maintain a cautious attitude toward irradiation sterilization processes.
After sufficient literature research, the author summarized the technical guidelines on irradiation sterilization at home and abroad, as shown in the following table:

In June 2014, PDA published a comprehensive review titled "Gamma Sterilization of Pharmaceuticals: A Review of Irradiation of Excipients, Active Pharmaceutical Ingredients, and Final Drug Product Formulations". The article provides detailed guidance on establishing irradiation conditions to ensure final product quality, while also introducing various quality control characterization methods for irradiated pharmaceuticals. These methods are used to evaluate structural integrity, active ingredient content, and the presence of degraded impurities in sterilized products.
3. Irradiation sterilization has a long way to go
Irradiation sterilization is influenced by multiple factors including microbial load, microbial population, moisture content, oxygen levels, environmental conditions, product density, irradiation dose, loading patterns, and radiation uniformity—all of which affect sterilization efficacy. This process impacts drug quality, packaging materials, and critical quality attributes of special formulations, requiring specialized methods to study both qualitative and quantitative changes. For products containing special carriers like polymer microspheres or nanoparticles, irradiation may cause cross-linking or chain breakage in macromolecular chains, necessitating analytical methods to determine maximum tolerable irradiation doses. Additionally, irradiation sterilization could induce radioactive decomposition in active pharmaceutical ingredients (APIs), potentially affecting product color, odor, stability, or bioactivity. Therefore, detailed studies on drug radiochemical stability are essential—such as evaluating product stability after maximum irradiation doses—to confirm sterilization's impact. These radiation-induced changes may influence product safety and efficacy, requiring thorough validation.
Current domestic and international guidelines on pharmaceutical irradiation sterilization remain fragmented and lack systematic organization. Notably, China's technical guidelines for irradiation sterilization primarily focus on traditional Chinese medicine (TCM), while chemical pharmaceuticals in this field remain underdeveloped. Regulatory agencies, industry associations, and research institutions should collaborate to establish regulatory frameworks through scientific studies and discussions, ensuring the method's scientific validity and practical applicability. Furthermore, it is imperative to expedite the development of standardized technical guidelines for both industry practitioners and regulators to adopt.
reference documentation
1、Gamma Sterilization of Pharmaceuticals—A Review of the Irradiation of Excipients, Active Pharmaceutical Ingredients, and Final Drug Product Formulations
2、EMA.Guideline on the sterilisation of the medicinal product, active substance, excipient and primary container
3. CDE. Technical Guidelines for Irradiation Sterilization of Traditional Chinese Medicine
Disclaimer: This article is only for the purpose of knowledge exchange and sharing, and does not involve commercial publicity, and does not serve as relevant medical guidance or medication advice. If there is any infringement, please contact us for deletion.
Our products are recommended:
1.1071-46-1 https://www.bicbiotech.com/product_detail.php?id=6342
2.113853-16-0 https://www.bicbiotech.com/product_detail.php?id=6343
3.2136-75-6 https://www.bicbiotech.com/product_detail.php?id=6351
4.98737-29-2 https://www.bicbiotech.com/product_detail.php?id=6352
5.1191-16-8 https://www.bicbiotech.com/product_detail.php?id=6353




