Introduction: In these turbulent years of biomedicine, the most common phrase you hear is "more rational".
Investment and financing is more rational, research and development is more rational, and commercialization is more rational.
Rationality is undoubtedly a positive force. Yet when it becomes "all about certainty," innovation
inevitably slows down – or even comes to a complete halt! A recent industry study sent a wake-up call: While biopharmaceutical companies are expanding their pipelines and securing more funding, the number of novel therapeutic targets entering R&D each year has plummeted to alarming levels.
From about 100 in the past year, it dropped to only 30 last year!
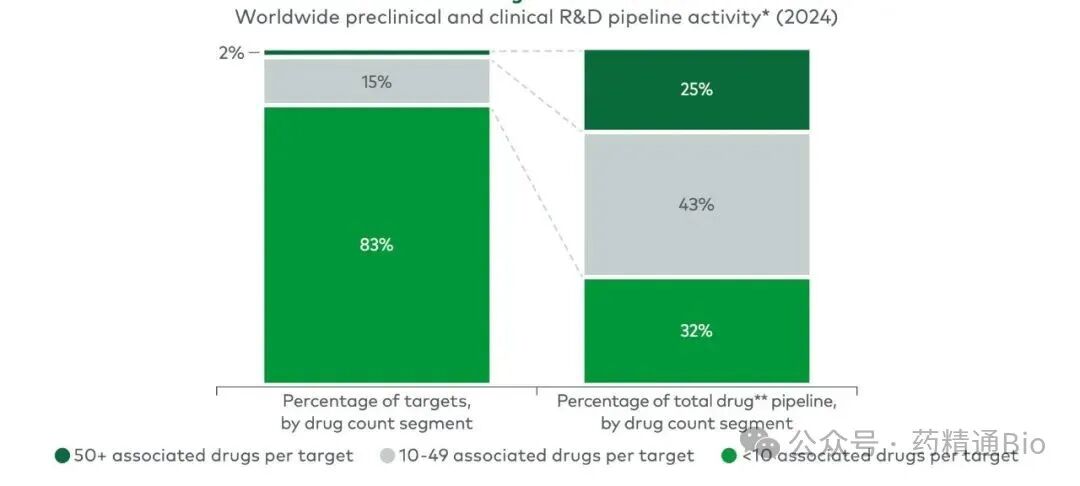
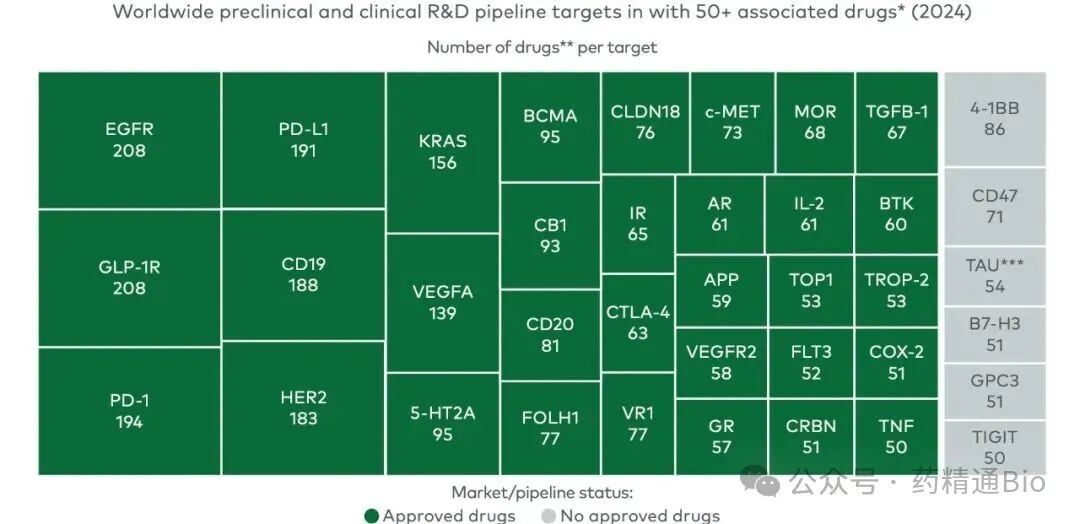
Meanwhile, a quarter of the industry's 13,600 drug-target pairings are concentrated on just 38 targets. Researchers are locked in a race to secure positions in highly predictable fields and targets
With so many scientists and companies worldwide competing, they ultimately cluster around dozens of familiar entry points.
Why? Because it is "ripe", and ripe is not easy to be wrong.
The problem is, the more familiar it is, the more crowded it is. To be afraid of risk does not mean to avoid risk!
1. The sense of security in the crowd: everyone goes to the same track
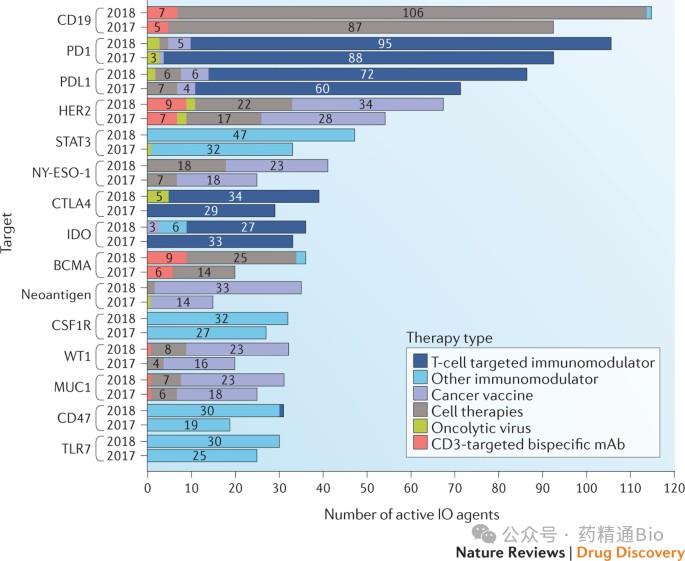
Imagine the proven track: GLP-1 (weight loss), PD-1/PD-L1 (tumor immunity), EGFR, HER2, KRAS, CD19/20, BCMA...
The industry's bigwigs are watching them.
Why? Because these targets have a more favorable risk profile: clear mechanisms, mature clinical pathways, regulatory familiarity, well-established biomarkers, and commercial validation.
As a result, industry innovation has increasingly embraced "micro-innovations": switching to different epitopes on the same protein; transforming small molecules into antibodies and antibodies into TCEs (target cell engineering) or adding delivery systems; adopting new strategies like precision stratification, combination therapies, and expanded indications. The list goes on!
Optimizing known target drugs is a good thing in itself to continue to generate value.
However, when too many resources are sucked into a few targeted drugs, the overall efficiency of the industry will decline: high repetition, difficult to differentiate, and the future of "crowded red sea" is felt from the beginning of project approval!
And it often takes a lot of money in the later phase 3 clinical, commercialization stage to go into the sea bubble bath!
Why is innovation slowing down
Many people would say that it is not less money, but that money is more "smart".
The data do show that the round A investment and financing has grown at a 10-year compound rate of about 18%, but the concentration has increased—— more money has been invested in fewer companies.
See: Biomedical industry financing: The logic has been restructured
The pipeline has doubled, from 11,000 in 2015 to 21,000 in 2024. But the pace of entry into "new targets" is slowing.
What does that mean? The money hasn't gone away, it's just become more "discerning."
How critical should the scrutiny be? When pitching a "first-in-class" target for funding, investors will ask: What's the likelihood of it becoming the "best in class"? Is the biomarker mature? How granular is the patient stratification? How well-defined is the clinical pathway? How many competitors are there? While these questions are valid, they often lack answers or sufficient clarity when evaluating "new targets".
As a result, resources are increasingly tilted toward "the second and third generations of known targets". This is a collective risk hedging, but it also delays the birth of "something truly new" in common.
Big opportunities are often in places where you can't see clearly
I particularly like the idea that the essence of innovation is to make "what is not clear" clear, rather than seeing it clearly from the beginning.
Over the past four years, about 350 new targets have entered development.
It focuses on six mechanisms: cell fate and differentiation, metabolism and clearance, enzymatic modification, immune cell balance, neuronal plasticity and activation, and protein degradation

About 70% of these directions remain in preclinical, and only 30% have entered phase I.
It's not that it's worthless, it's that it's still in the "unclear" phase.
Instead of "avoiding", they need to be smarter at "de-risking": using better models, data and experimental designs to peel away layers of uncertainty.
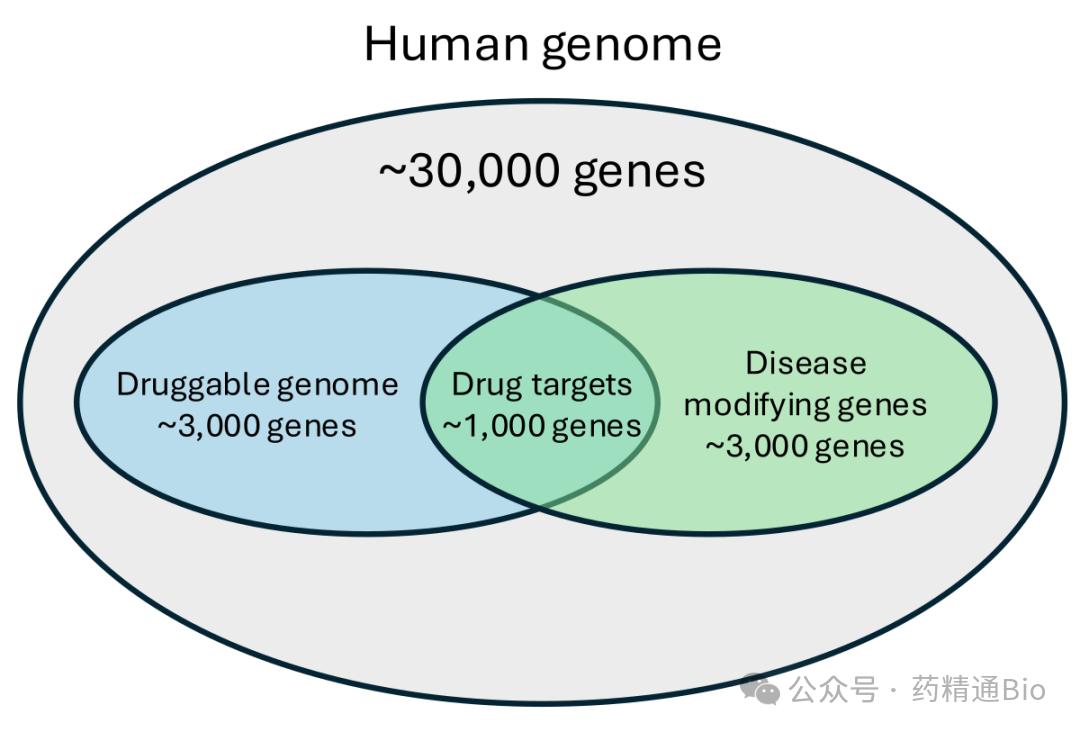
Why is it worth doing?
Because more than half of the human proteins that can be made into drugs have never been touched by drugs. In other words, the surface is busy and the bottom is quiet.
The real blue ocean is still below.
4. Find relative certainty in uncertainty
In business, we should be "strategically clear and tactically flexible", which is also true for new targets of innovative drugs.
When many companies make annual plans, they only set an "ROI threshold" but not a "novelty threshold".
In fact, we should set a clear proportion suitable for our company for the first mechanism, such as 20% -30% of "original exploratory expenditure", and do not take short-term milestones as the only standard for project assessment.
Without leaving room for "exploration without certainty", organizations are pushed by KPIs toward "repetition of certainty".
However, recent years have seen significant progress. Previously, many critical hypotheses could only be validated through Phase I clinical trials. Now, with technologies like AI, humanized systems, organoids, and in situ multi-omics, we can obtain efficacy and toxicity data
earlier, dramatically improving the success rate of FIH (First-in-Human) trials. Moreover, many projects expand their efforts in a scattergun approach, only considering differentiated competitive strategies when they vaguely reach Phase II – by then it's already too late.
A "differentiation blueprint" should be made at the beginning of project initiation and drug discovery: target biology, molecular mode, delivery system, combination, patient stratification, endpoint selection, and how each link forms a barrier. The "technology roadmap" should be turned into a "competition roadmap".
Don't turn "rationality" into "conservatism"
Innovation is not about "escaping" risk, but about dancing with it. There is no need to play hard to get, but you should not bet all your resources on the "comfort zone".
There is a paradox in the business world: in the short run, it is safest to choose certainty; in the long run, it is most dangerous to choose certainty. Because certainty is the easiest to crowd, and crowding is the easiest to be mediocre.
The essence of strategy is to find relative certainty in uncertainty. What would you do if you were in charge of a company's pipelines?
Are most of your pipelines in the top 30-40 targets that are most crowded in the industry? What is the annual budget for innovation?
I hope we can not make "rationality" into "conservatism";
Replace the sense of security with a sense of certainty about the future.
Real innovation always happens where it's not clear!
Disclaimer: This article is only for the purpose of knowledge exchange and sharing, and does not involve commercial publicity, and does not serve as relevant medical guidance or medication advice. If there is any infringement, please contact us for deletion.
Our products are recommended:
1.866594-60-7 https://www.bicbiotech.com/product_detail.php?id=6364
2.82657-04-3 https://www.bicbiotech.com/product_detail.php?id=6365
3.115852-48-7 https://www.bicbiotech.com/product_detail.php?id=6366
4.83-33-0 https://www.bicbiotech.com/product_detail.php?id=6367
5.104075-48-1 https://www.bicbiotech.com/product_detail.php?id=6368




