Once a "sky-high priced cancer drug" costing millions of yuan, it is quietly initiating a revolution concerning life and accessibility. On November 7, 2025, "Purikiorunase Injection" developed by Chongqing Precision Biotechnology Co., Ltd. was approved for market release by the National Medical Products Administration, becoming China's first CAR-T cell therapy targeting refractory or relapsed B-cell acute lymphoblastic leukemia in children and adolescents. Just days before this product's approval, media reports indicated that the 2025 National Medical Insurance Negotiations would for the first time explore introducing the "Commercial Insurance Innovative Drug Catalog," with multiple CAR-T therapies expected to enhance accessibility through this initiative.
From the approval of China's first CAR-T product in 2021, it has only taken four years for China's CAR-T therapy to enter a fast track. Currently, eight products have been approved for market, covering various hematologic tumor indications, and are gradually transitioning from high-end customization to a new phase of inclusive healthcare.
1. Comprehensive Analysis of Domestic CAR-T Products on the Market
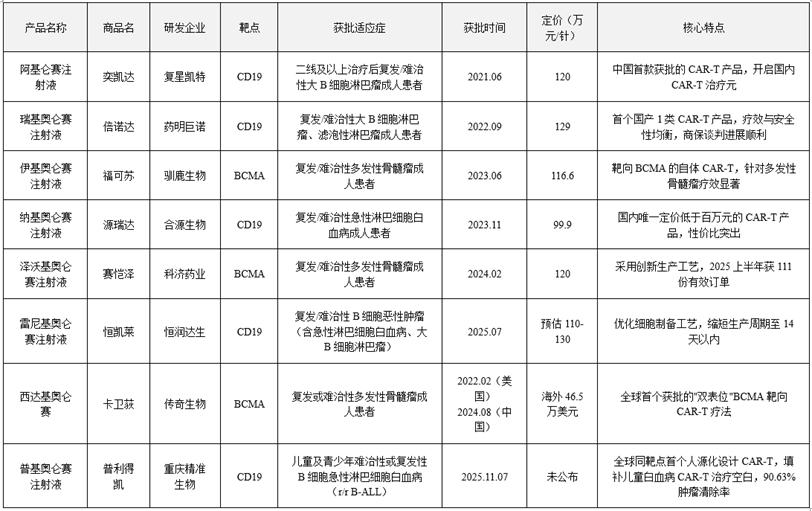
2. Core Features of Current CAR-T Products
1. Target Concentration, Focus on the Blood Tumor Needs
The eight approved CAR-T products demonstrate highly concentrated therapeutic targets, with five targeting CD19 and three targeting BCMA, covering common hematologic malignancies including relapsed/refractory large B-cell lymphoma, acute lymphoblastic leukemia, and multiple myeloma. This characteristic is closely tied to the current maturity of CAR-T technology. CD19 and BCMA targets exhibit high surface expression on hematologic tumor cells with low immunogenicity, making them the most ideal CAR-T therapeutic targets for achieving precise tumor cell targeting.
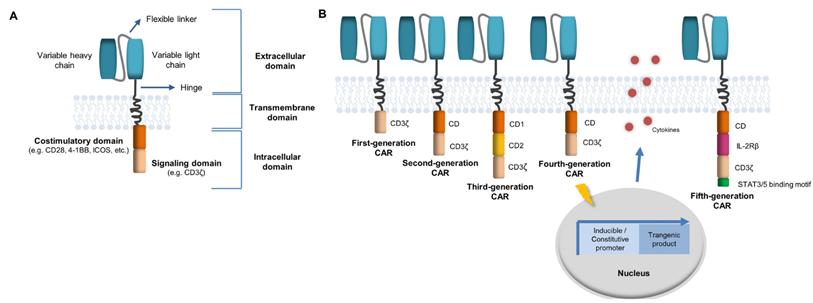
Figure 1: CAR architecture (second generation) and its evolution to fifth-generation CAR technology (Reference 9)
2. The technical approach prioritizes autologous CAR-T therapy, with some products achieving innovative breakthroughs.
All currently available products are autologous CAR-T therapies, which follow a 'one patient, one batch' customization model. The process involves harvesting T cells from the patient's body, genetically modifying and expanding them in vitro, before reinfusing them into the patient.
3. Prices generally exceed one million, and the payment threshold remains high
CAR-T products are typically priced between 1 million and 1.3 million yuan per dose. Heshen Biotech's Nekerolimab, however, is priced at 999,000 yuan per dose, just below the 1 million yuan threshold. The high cost stems from its "customized production" approach—each patient's cells require individual cultivation and testing, with a production cycle of 3-4 weeks and substantial R&D investment, resulting in persistently high unit costs. This price makes the therapy unaffordable for most families and limits its widespread adoption.
4. Expanded from adults to children, to meet the needs of special populations
Early CAR-T therapies were primarily designed for adult patients. With the approval of Pemucizumab by Chongqing Precision Biotech, the treatment has now expanded to pediatric and adolescent populations, filling a critical gap in pediatric leukemia CAR-T therapy. Notably, in pivotal Phase II clinical trials, Pemucizumab achieved a 90.63% (58/64) optimal complete response/complete response with incomplete recovery (CR/CRi) rate within three months, including 78.13% (50/64) complete remission (CR) and 12.5% achieving complete remission with incomplete blood cell recovery (CRi). This breakthrough provides a new therapeutic option for pediatric and adolescent patients.
III. Breaking the Payment Barrier: From Patient Out-of-Pocket to Multi-Payment System
While CAR-T therapy delivers remarkable efficacy, its exorbitant costs—often reaching millions of yuan—have long hindered its widespread adoption for average families. Now, through policy innovations and market exploration, a diversified payment ecosystem is taking shape.
China's First Commercial Insurance Innovative Drug Catalogue Negotiated, Five Products Expected to Benefit
In 2025, China's national medical insurance negotiations officially introduced the "Commercial Insurance Innovative Drug Catalog" mechanism for the first time. Five CAR-T therapies passed expert review for inclusion in this catalog: Nekyolunsi by Heyuan Biotech, Ijiolunsi by Xunlu Biotech, Ajiolunsi by Fosun Kite, Ruijiolunsi by WuXi JUNO, and Zewoqilunsi by Kojie Pharma. Notably, three of these therapies have been submitted simultaneously to both the National Basic Medical Insurance Catalog and the Commercial Insurance Innovative Drug Catalog, which is expected to reduce patient costs through the combined "medical insurance + commercial insurance" model.
2. Commercial insurance and people's insurance are laid out in advance, and the coverage is gradually expanded
Prior to the establishment of China's commercial insurance drug catalog mechanism, the market had already developed a preliminary multi-payment structure. By the end of 2024, Fosun Kite's Aclimutinib had been included in over 80 commercial medical insurance plans, while WuXi JUNO's Relinucel was covered by approximately 70 commercial insurance products. In the Huiminbao (People's Welfare Insurance) program, more than 60% of its 2024 coverage items included CAR-T therapies, laying the groundwork for the official launch of the commercial insurance catalog. This hybrid model of "commercial insurance + Huiminbao" has effectively alleviated medication costs for many patients.
3. Payment Mode Innovation and Sustainable Path Exploration
The payment mechanisms for CAR-T therapies are currently exploring various innovative models, including "pay-for-performance," "installment payments," and "public welfare assistance." Some pharmaceutical companies have partnered with charitable organizations to launch patient aid programs, offering medication grants or cost reductions to eligible patients. Certain commercial insurance products adopt models such as "treatment first, payment later" or "payment upon achieving treatment targets," thereby reducing treatment risks for patients. With the implementation of the commercial insurance innovative drug catalog, a multi-tiered payment system is expected to emerge, featuring "basic medical insurance coverage, commercial insurance supplements, and public welfare assistance," significantly enhancing the accessibility of CAR-T therapies.
IV. Technological Innovation: Three Development Tracks of Next-Generation CAR-T
While existing CAR-T products have demonstrated remarkable efficacy in hematologic cancer treatment, challenges such as "customized production, high costs, and lengthy preparation cycles" remain unresolved. Scientists and pharmaceutical companies are actively exploring next-generation CAR-T technologies, primarily focusing on three key areas:
1. Spot CAR-T (UCAR-T): Industrialization reduces costs
Spot CAR-T technology isolates T lymphocytes from healthy donors and employs gene-editing techniques to eliminate genes associated with graft-versus-host disease and immune rejection. This approach enables industrial-scale production under GMP standards, with frozen storage allowing immediate patient administration without preparation cycles. Compared to autologous CAR-T, spot CAR-T production costs can be reduced by over 80%, fundamentally transforming the "one-in-a-million" pricing model. Currently, Koj Pharmaceutical's generic BCMA CAR-T product CT0596 and CD19/CD20 CAR-T product CT1190B, developed on the THANK-u Plus® platform, have demonstrated promising clinical data showing favorable safety and efficacy profiles. The first generic CAR-T product is expected to hit the market within 4-5 years.
2. In vivo CAR-T: Simplifying the Treatment Process
In vivo CAR-T therapy enables direct delivery of CAR-coding genes to T cells within the patient's body, facilitating in situ generation of CAR-T cells. This approach eliminates the need for ex vivo expansion and reinfusion, significantly streamlining the treatment process and shortening preparation cycles. According to Novotech's 2025 industry report, over 70 global in vivo CAR-T research projects were underway in 2024, with projections indicating a milestone of 100 projects by 2025. Of these, 75% are in preclinical stages, while 5 have progressed to clinical trials. This "one-shot" approach not only reduces costs but also benefits patients who cannot tolerate cell harvesting, representing a crucial direction for the widespread adoption of CAR-T therapy.
3. New Target Expansion and Breakthrough in Solid Tumors: Breaking the Treatment Boundary
Currently, CAR-T has achieved success in the treatment of hematologic tumors, but it still faces challenges in the treatment of solid tumors. The China research team has made breakthrough progress. The "nanobody-armored CAR-T cells (NAC-T)" developed by institutions such as Shanghai University Affiliated Mengchao Cancer Hospital successfully treated malignant mesothelioma by equipping CAR-T cells with "anti-PD-1 nanobody armor." In the Phase I clinical trial, 11 patients who failed standard treatment achieved a 100% disease control rate after treatment, with 63.6% of patients showing significant tumor shrinkage. The median overall survival reached 25.6 months, far exceeding the current second-line treatment level of approximately 11 months, and one patient has survived tumor-free for over 3.5 years. Additionally, Kojii Pharmaceutical's Shuruiji Orelumab Injection (targeting Claudin18.2) has submitted a new drug marketing application for the treatment of advanced gastric/oesophageal gastroesophageal junction adenocarcinoma, which is expected to become the first CAR-T product targeting solid tumors.
V. Future Outlook: From High-end Customization to Universal Healthcare
The future of China CAR-T therapy will revolve around three major directions: technological breakthroughs, payment innovations, and expanded indications, ultimately achieving a transition from high-end customization to universal healthcare.
From a technical standpoint, spot-type CAR-T and in vivo CAR-T are poised to become the dominant therapeutic approaches. With advancements in gene-editing technology and optimized manufacturing processes, CAR-T production costs are projected to drop significantly. Within the next 5-10 years, CAR-T therapy pricing is expected to fall below 300,000 yuan, making it accessible to average households. Meanwhile, breakthroughs in solid tumor CAR-T will expand treatment options to cover common cancers like lung, liver, and stomach cancer, benefiting a broader patient population.
In terms of payment mechanisms, the introduction of the "Commercial Insurance Innovative Drug Catalog" in 2025 will mark a pivotal turning point. With more CAR-T products being included in both commercial insurance and medical insurance catalogs, combined with the extensive coverage of the People's Welfare Insurance, a multi-tiered payment system will be established, significantly reducing patients' out-of-pocket expenses. By 2027, it is projected that the out-of-pocket costs for domestic CAR-T patients will drop below 30% of total treatment expenses, substantially improving accessibility.
At the industrial level, China's CAR-T industry is transitioning from following to running side by side and even leading the way. Chongqing Precision Biotech's Pemigolizumab, as the world's first humanized CAR-T product targeting the same site, has provided a "China solution" for global pediatric leukemia treatment; companies such as Koj Pharmaceutical and Xunlu Biotech have made research and development progress in general CAR-T and solid tumor CAR-T that are among the world's top tier. With more innovative products approved and technology exported, China is expected to become a global innovation hub for CAR-T therapy.
From the "sky-high priced drugs" costing millions per dose to the gradually popularized "lifesaving drugs", the development journey of China's CAR-T therapy is a microcosm of China's biomedical innovation. With continuous technological breakthroughs, increasingly improved payment systems, and expanding indications, CAR-T therapy will truly enter ordinary households, enabling more cancer patients to benefit from this revolutionary treatment and ushering in an era of inclusive cancer treatment.
reference material
1. https://mp.weixin.qq.com/s/z0ZgRMdG8cQP_paBma1Gww
2. http://m.toutiao.com/group/7569406341312217652/?upstream_bizdoubao
3. http://m.toutiao.com/group/7569777043232899624/?upstream_bizdoubao
4. http://www.cqnews.net/1/detail/1436396511222067200/app/content_1436396511222067200.html
5. http://m.toutiao.com/group/7569196554964288006/?upstream_bizdoubao
6. http://m.toutiao.com/group/7567267342707704363/?upstream_bizdoubao
7. http://m.toutiao.com/group/7567378238943527476/?upstream_bizdoubao
8.https://m.nbd.com.cn/articles/2025-11-02/4126918.html
9. Current challenges and emerging opportunities of CAR-T cell therapies
10. Anti-PD-1 Nanobody-Armored MSLN CAR-T Therapy for Malignant Mesothelioma: Preclinical and Clinical Studies
Disclaimer: This article is for knowledge exchange, sharing, and educational purposes only. It does not constitute commercial promotion, medical advice, or medication recommendations. If the content infringes on any rights, please contact us for removal.
Our product recommendations:
1.2719749-02-5 https://www.bicbiotech.com/product_detail.php?id=6444
2.1227502-89-7 https://www.bicbiotech.com/product_detail.php?id=6445
3.1227593-77-2 https://www.bicbiotech.com/product_detail.php?id=6446
4.2185857-97-8 https://www.bicbiotech.com/product_detail.php?id=6447
5.101303-98-4 https://www.bicbiotech.com/product_detail.php?id=6448




