Author: Aoyama
For an extended period, the immunology and inflammation drug market has been divided by a seemingly insurmountable divide: on one side are small-molecule targeted agents, which offer the significant convenience of oral administration and the ability to penetrate intracellular targets, but are often limited by off-target toxicity and efficacy ceilings in large-area PPI interactions; on the other side are large-molecule biologics, such as monoclonal antibodies, which exhibit high specificity and potent therapeutic effects, yet must rely on injectable administration, imposing compliance burdens on patients and incurring cold chain transportation costs.
However, the advent of the oral macrocyclic peptide JNJ-2113 (Icotrokinra) marked the arrival of a new era in oral biologics. Through macrocyclization technology, JNJ-2113 successfully circumvented the limitations of the traditional five rules of drug design, achieving both high affinity and selectivity at the antibody level while demonstrating sufficient oral bioavailability to produce systemic therapeutic effects.
01. Breaking the deadlock: from the forgotten middle ground
In the textbooks of medicinal chemistry, there is a renowned Rule of 5 by Ribinsky, which stipulates that oral small molecules must possess appropriate molecular weights, hydrogen bond donors/acceptors, and lipophilic/hydrophobic distribution. This rule acts like a curse, restricting the molecular weight and hydrophilicity/hydrophobicity of oral drugs. For a long time, polypeptides with molecular weights in the 500–2000 Da range have often exhibited significant conformational flexibility in solution, leading to substantial conformational entropy costs during target binding. Meanwhile, their molecular size and polar characteristics limit passive absorption through the intestinal epithelium, keeping them in a gray zone between small and large molecules in oral drug development.
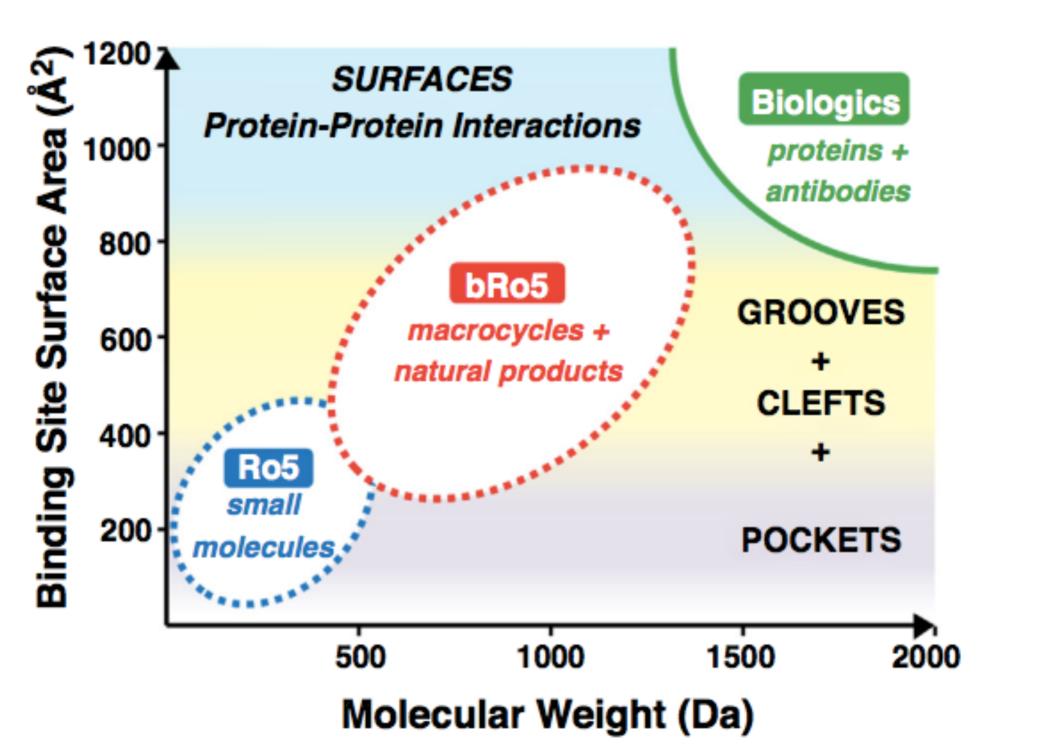
However, with JNJ-2113, co-developed by Johnson & Johnson and Protagonist Therapeutics, demonstrating dominant efficacy and safety data in Phase III clinical trials for psoriasis, this previously considered insurmountable molecular scale range has begun to reveal unprecedented development potential. As the world's first orally administered IL-23 receptor antagonist peptide to successfully advance to late-stage clinical trials, JNJ-2113 is challenging an industry consensus: Who says that achieving biologic-level efficacy requires enduring the pain of injections?
02. R&D: A Decade of Refinement, The Evolutionary History of JNJ-2113
JNJ-2113 did not emerge out of nowhere; it is the result of iterative evolution from Protagonist's peptide technology platform. Scientists at Protagonist utilized their proprietary Vectrix™ platform to establish a vast library of disulfide-rich peptides (DRPs). These peptides mimic the highly stable molecular structures found in nature, such as snake venom and anemone toxins. Unlike linear peptides, which are conformationally unstable, DRPs resemble pre-organized micro-antibodies that require no significant conformational changes to bind to targets, thereby ensuring high affinity.
The predecessor of JNJ-2113 was PTG-200. Initially, when Johnson & Johnson and Protagonist entered into a $990 million collaboration agreement in 2017, the objective was not systemic administration but rather the development of an intestinal-restricted therapy.
The rationale of PTG-200: Given the poor oral absorption of peptides, it is logical to design the drug to remain exclusively in the intestinal tract for the targeted treatment of inflammatory bowel disease (IBD). This approach enables the utilization of high-concentration drugs to directly act on the lesion while avoiding systemic immunosuppressive side effects associated with parenteral administration.
The Evolution of JNJ-2113: As research progressed, the team uncovered greater potential in the Vectrix™ technology platform—achieving maximum affinity could enable sufficient therapeutic efficacy even with minimal drug exposure. This led to a significant shift in R&D strategy: from exploiting non-absorption to overcoming it. Building upon PTG-200, Johnson & Johnson and Protagonist's medicinal chemistry teams optimized affinity, stability, and systemic activity, ultimately enhancing JNJ-2113's affinity by over 1,000-fold. Its Kd reached 7.1 pM, comparable to that of antibodies. In experiments inhibiting IL-23-induced STAT3 phosphorylation, the IC50 was 5.6 pM, while in whole blood IFN-γ release assays, the IC50 was 9 pM, demonstrating exceptional in vitro activity.
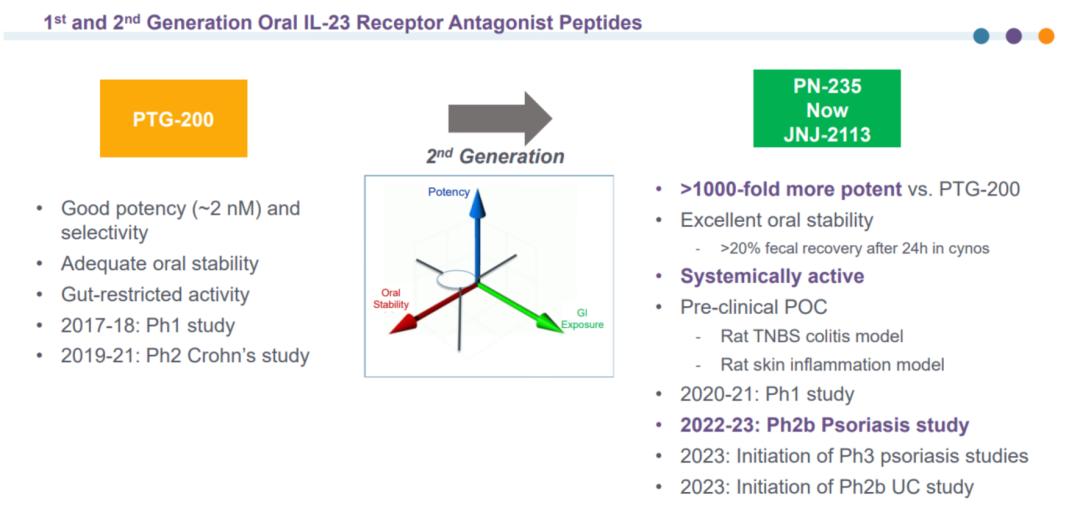
Evolution of PTG-200 to JNJ-2113
Beyond efficiency enhancement, the R&D team faces a core challenge: how to design a peptide with a molecular weight of 1500-2000 that can withstand the attack of gastric acid and proteases while penetrating the dense intestinal epithelial cells to enter the bloodstream?
macrocyclic rigid skeleton
JNJ-2113 is not merely a peptide but a macrocyclic structure. Linear peptides, upon entering the gastrointestinal tract, are rapidly degraded by trypsin and chymotrypsin due to thermal motion exposing various enzymatic cleavage sites, akin to noodles being shredded. In contrast, JNJ-2113 forms a rigid, compact cyclic structure by linking peptide chains end-to-end via disulfide bonds or introducing internal constraints. This rigid structure locks the molecular active conformation while concealing cleavage sites such as amide bonds, endowing it with exceptional metabolic stability in the harsh gastrointestinal environment. After 24 hours of treatment with various gastrointestinal proteases, 84%-100% of the molecules remain unmetabolized, whereas only 3% of JNJ-2113 is degraded after 2-hour incubation in hepatocytes.
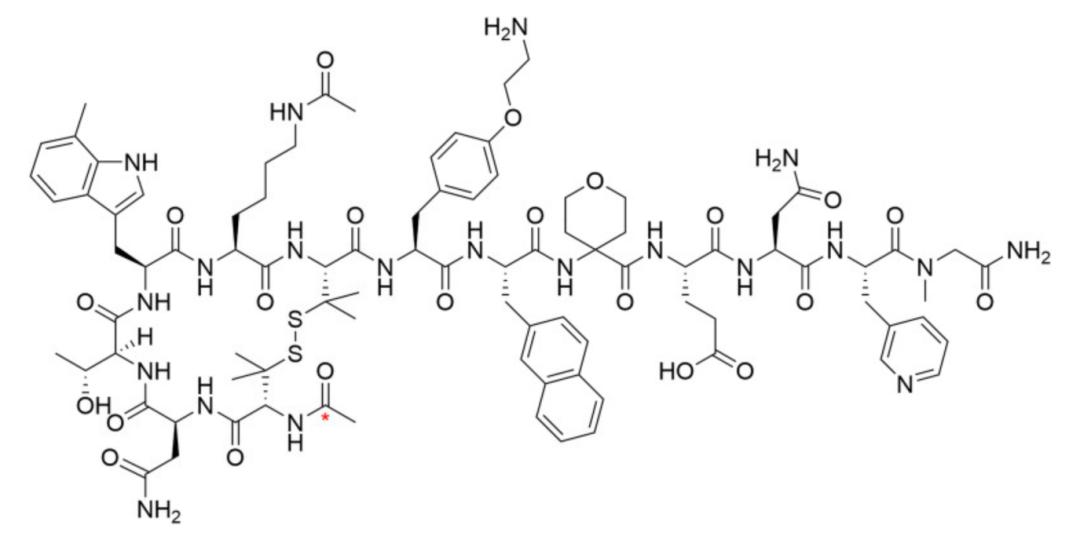
Molecular structure of JNJ-2113 (MW: 1898.17)
Use of osmotic agents
Enhancing peptide stability often reduces its membrane permeability. JNJ-2113 achieves this by employing sophisticated side-chain modifications and macrocyclic structures that partially mask polar surfaces like peptide bonds while maintaining water solubility, thereby lowering the overall desolvation energy barrier. This property enables the cyclic peptide to more effectively cross biological barriers such as cell membranes or intestinal mucosa, facilitating certain oral bioabsorption. Additionally, JNJ-2113 utilizes oral pro-membrane technology in its formulation, incorporating amphiphilic molecules like sodium decanoate and SNAC. These excipients can transiently modulate the fluidity of epithelial cell membranes or briefly open tight junctions in the stomach or small intestine, facilitating the passage of macrocyclic peptide molecules and enhancing JNJ-2113's penetration of the intestinal mucosa.
According to the preclinical data of JNJ-2113, its F% in rats and monkeys was approximately 0.3%. However, due to its strong molecular activity and long half-life, even at a low plasma concentration (Cmax of 84 ng/mL at a dose of 22.5 μMpk), it could maintain prolonged effective inhibition. The IC50 values for STAT3 and IFN-γ were 10–18 ng/mL.
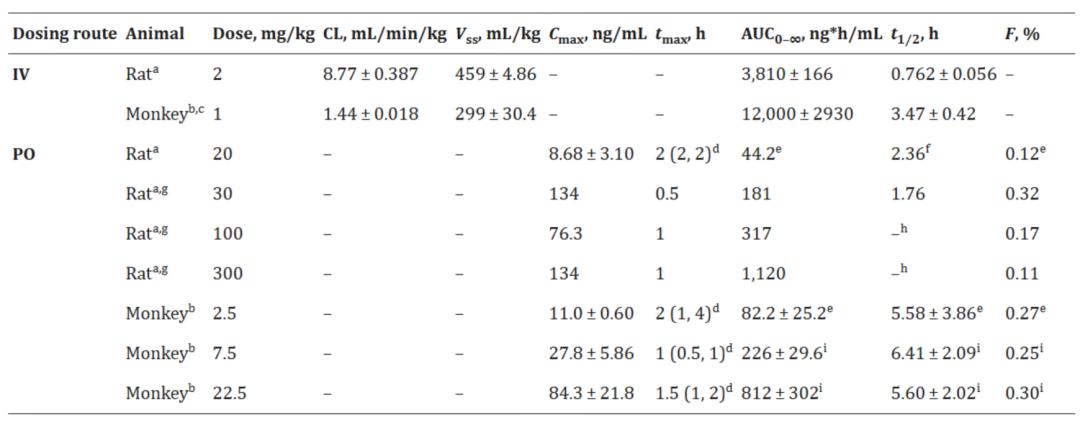
Animal PK of JNJ-2113
03.Tripartite Conference on Clinical Data
In the highly competitive market of autoimmune diseases, JNJ-2113 faces a dual challenge: the threat of antibodies (wolves ahead) and the dominance of oral standard therapy (Sotyktu behind). While Skyrizi (IL23 antibody) and Stelara (IL12 antibody) demonstrate favorable efficacy, they require injection, whereas Sotyktu, though administered orally, exhibits significantly inferior safety and efficacy compared to antibody-based therapies. From its inception, JNJ-2113 was designed to integrate the advantages of both approaches, aiming to achieve truly oral-grade antibody efficacy.
According to the latest published Phase III data, JNJ-2113 demonstrated significantly superior efficacy to the standard oral treatment Sotyktu in the treatment of moderate to severe psoriasis. The response rate (IGA0/1) reached 70% at 16 weeks, compared to 54% for Sotyktu; the PASI100 score at 16 weeks was 32%, which was close to the historical data of Skyrizi and more than twice that of Sotyktu. No new safety signals were observed, with the most common adverse events being nasopharyngitis (6% vs 5% vs 9%) and upper respiratory tract infections (4% vs 3% vs 5%). In terms of serious adverse event (SAE) incidence, JNJ-2113 showed a rate of 2.8%, comparable to Skyrizi and Sotyktu, with no dose-limiting toxicity observed. This demonstrates that macrocyclic peptides successfully avoid off-target risks associated with traditional small molecules while maintaining high activity.
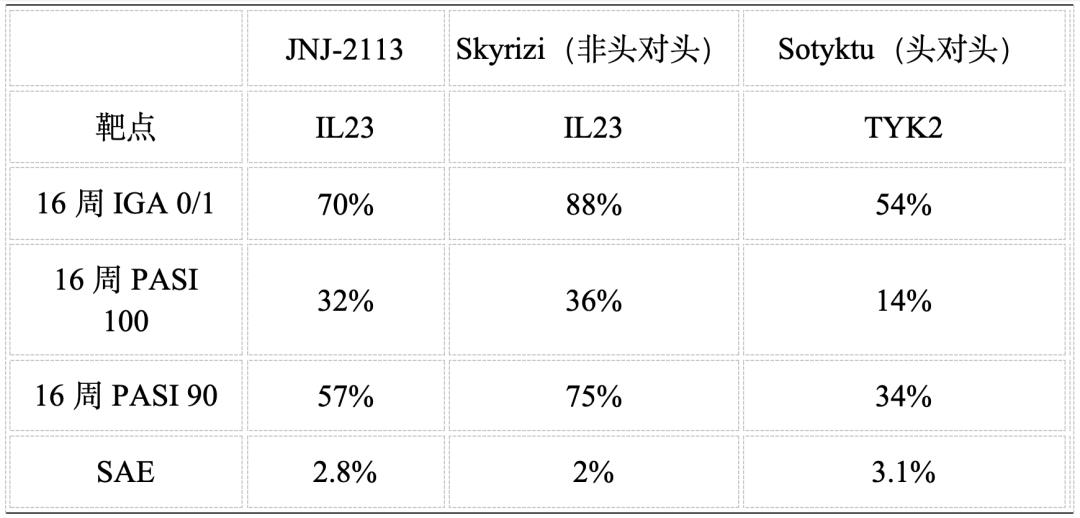
Comparison of Therapeutic Efficacy in Moderate to Severe Psoriasis
JNJ-2113 can be said to have completely outperformed Sotyktu, and its 16-week PASI 100 response rate has approached that of Skyrizi, thoroughly dispelling the stereotype that small-molecule oral drugs, despite their efficacy, are inferior to biologics. For patients weary of injections, the trade-off of slightly lower absolute efficacy for needle-free administration represents an exceptionally attractive option. Admittedly, JNJ-2113 still lags slightly behind Skyrizi in absolute numerical terms, but it has become sufficient to challenge the market dominance of first-generation biologics such as Humira and Stelara, establishing itself as the preferred choice for needle-phobic patients. Moreover, this may only be the "first-generation" of macrocyclic peptides. With advancements in phage display and chemical modification technologies, the singularity of oral biologics has arrived. The gap with Skyrizi may no longer be an insurmountable divide, and it is reasonable to believe that the emergence of orally administered peptides equivalent to antibodies is only a matter of time.
04. Identification of the Next Macrocyclic Peptide
JNJ-2113 has demonstrated that oral macrocyclic peptides represent a clinically successful modality, combining the high activity and selectivity of antibody drugs with the lower production costs of chemical drugs, while also supporting oral administration. This implies that companies possessing peptide oral formulation technologies, macrocyclic peptide library screening platforms, or phage display technologies will see further elevation in the value of their pipelines. Given Johnson & Johnson's strong commercialization capabilities, subsequent entrants targeting IL-23 oral peptides as mere Me-too products will have no opportunity. Instead, more opportunities lie in:
·Extracellular Target Expansion: Utilizing the same platform technology to address other validated targets (e.g., oral administration of IL-17 and TNF-alpha). Protagonist has initiated Phase I clinical trials for PN-881 (a potential BIC oral IL-17 antagonist), while Merck's MK-0616 (a PCSK9 inhibitor) aims to introduce oral macrocyclic peptides into the trillion-dollar cardiovascular chronic disease market, with Phase III clinical trials already achieving success.
·Intracellular target development: By developing cell-penetrating peptides with enhanced membrane permeability and leveraging their larger target contact area, this approach effectively blocks intracellular protein-protein interaction (PPI) targets (e.g., KRAS, MYC) that are challenging to drug develop with small molecules, offering high-risk, high-reward potential.
In conclusion
The story of JNJ-2113 is not only another triumph for Johnson & Johnson in the field of autoimmune diseases, but also a watershed moment in the history of drug development. It demonstrates that the route of administration is no longer a label distinguishing therapeutic efficacy. As the 'king of drugs,' Humira gradually ages, and small-molecule JAK inhibitors still struggle with safety concerns, JNJ-2113 is ushering in the next decade with its perfect trinity of antibody-level efficacy, oral convenience, and exceptional safety.
For patients with psoriasis, this means that taking a single tablet in the morning can achieve the improvement of skin lesions that previously required outpatient antibody injections. For drug development, it may be possible to re-examine all antibody targets through oral administration.
Ref:
1.https://www.galchimia.com/twenty-years-of-the-rule-of-five/
2.https://irp.cdn-website.com/
3.Sci Rep. 2024 Jul 30;14(1):17515.
4.https://mp.weixin.qq.com/s/SrtV3Y9LsNV-ZyCyn_esVg
5.Dermatol Ther (Heidelb). 2025 Sep;15(9):2495-2520
6.Lancet. 2025 Sep 27;406(10510):1363-1374.
7.https://www.skyrizi.com/psoriasis/psoriasis-treatment/skyrizi-results
8.https://doi.org/10.3389/fnut.2024.1496706
Disclaimer: This article is intended solely for knowledge exchange, sharing, and popular science purposes, and does not constitute commercial promotion, nor should it be regarded as medical guidance or medication advice. For copyright infringement, please contact us for removal.
Our product recommendations:
1.180864-33-9 https://www.bicbiotech.com/product_detail.php?id=6512
2.41307-63-5 https://www.bicbiotech.com/product_detail.php?id=6513
3.88909-96-0 https://www.bicbiotech.com/product_detail.php?id=6514
4.51411-04-2 https://www.bicbiotech.com/product_detail.php?id=6515
5.64369-13-7 https://www.bicbiotech.com/product_detail.php?id=6516




