In October 2025, Novartis agreed to acquire Avidity Biosciences for $12 billion, a move that drew public attention to a novel therapeutic model rapidly advancing through clinical trials: antibody-oligonucleotide conjugates. The Swiss pharmaceutical giant made generous offers for three late-stage AOC products, which not only constituted the second-largest transaction of 2025 but also marked growing industry interest in a platform capable of delivering oligonucleotides with antibody-like precision and eliciting patient responses surpassing those of existing therapies.
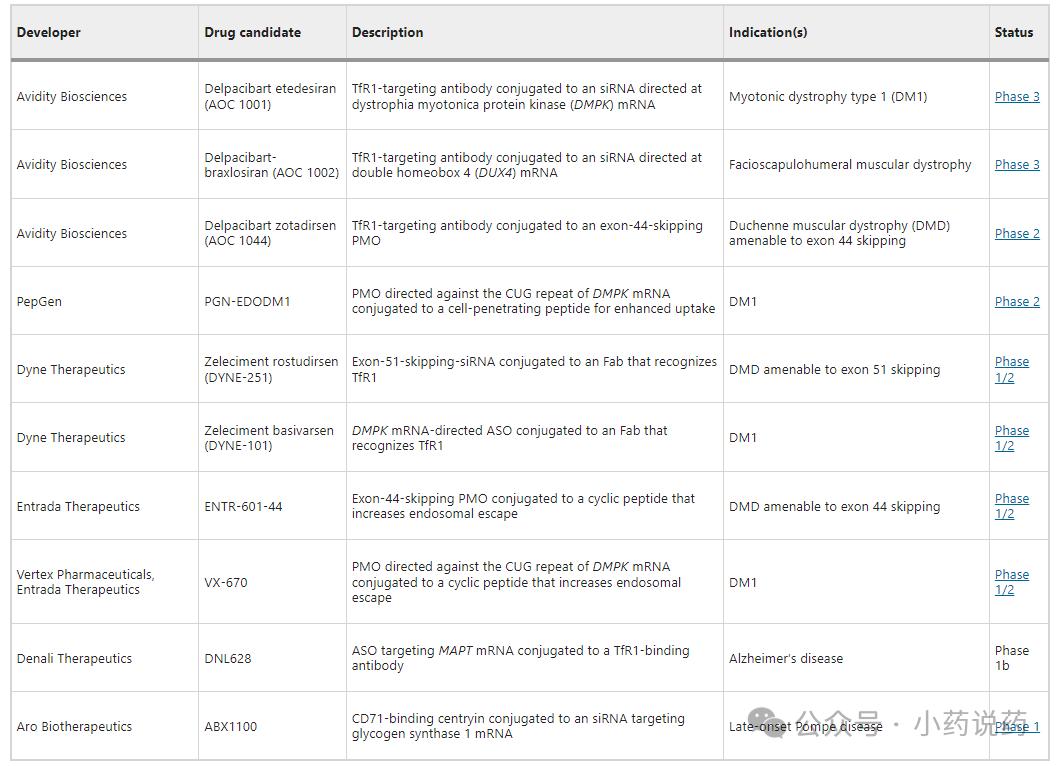
AOC is a drug model that combines gene regulatory oligonucleotides with the targeted precision of antibodies. Leading in this field are Avidity Biosciences and Dyne Therapeutics, both of which are developing antisense drugs delivered to skeletal muscle via transferrin receptor 1 (TfR1) for the treatment of Duchenne muscular dystrophy (DMD). Avidity's delpacibart zotadirsen (abbreviated as "del-zota") is an antibody-conjugated exon skipping antisense drug currently in Phase 3 clinical trials, with a Biologics License Application (BLA) pending submission. Meanwhile, Dyne Therapeutics is developing a exon skipping drug named zeleciment rostudirsen (abbreviated as "z-rostudirsen," DYNE-251) for the treatment of another form of DMD, with plans to submit a BLA in the second quarter. If approved, these novel combination therapies will influence gene expression at efficacy levels not observed with unbound drugs.
Interest in AOCs has surged as companies recognize the potential of combined drug entities to modulate gene expression with the flexibility of therapeutic oligonucleotides while achieving cell-specific targeting with the precision of antibodies. The overarching goal is to target any given tissue, including the central nervous system. However, the exogenous delivery of small interfering RNAs (siRNAs) and antisense oligonucleotides remains far from a well-established scientific approach, with significant room for development in creating tissue-specific drug shuttles and optimizing numerous performance parameters associated with these powerful yet complex molecules.
AOC shares many similarities with antibody-drug conjugates (ADCs), which are widely used as cancer therapeutic agents: both contain therapeutic payloads and targeting elements, and are linked through linkers. The primary distinction lies in the fact that ADCs typically carry non-specific cytotoxic payloads and are conjugated with antibodies targeting highly expressed tumor antigens (e.g., HER2 and TROP2), whereas the receptors recognized by AOCs are generally unrelated to the pathology targeted by the drug. However, AOCs require high expression at target tissues and must undergo rapid recirculation following endocytosis and AOC release.
Clinical data demonstrate that AOC molecules can significantly enhance the efficacy of existing therapies. For instance, Dyne's z-rostudirsen exhibited superior therapeutic outcomes in clinical trials compared to Exondys 51, an unbound antisense drug approved by Sarepta Therapeutics. Data revealed that muscle dystrophy protein levels in patients treated with rostudirsen increased from baseline normal values of 0.83% to 5.46% after six months, representing nearly a sevenfold growth. Meanwhile, Avidity's DMD drug del-zota elevated average dystrophin production to approximately 30% of normal levels in early clinical trials, which may have transformative implications for numerous patients.
In addition to DMD, Avidity and Dyne also have TfR1-mediated delivery programs for type 1 dystrophism (DM1) and facioscapulohumeral dystrophy. Notably, unlike Avidity, Dyne opted for a Fab antibody fragment targeting TfR1 rather than a full-length antibody, as the reduced size of the construct would enhance penetration in muscle tissue and the central nervous system. Furthermore, Ionis Pharmaceuticals is utilizing small TfR1 binders to deliver oligonucleotide drugs to target tissues and has observed "encouraging results" in transporting drugs across the blood-brain barrier using specific peptides.
However, the widespread expression of TfR1 across multiple tissues indicates its incomplete specificity, posing challenges such as reticulocytes potentially serving as a "reservoir" for drug delivery doses. To address this issue, Manifold Bio aims to identify alternative receptors to provide more tissue-specific pathways for drug delivery. The company has developed a screening method capable of delivering therapeutic payloads to the central nervous system of animal models with five times higher efficiency compared to TfR1-based strategies. This innovative approach has attracted interest from Roche, which has paid substantial fees to acquire access to these novel drug shuttles.
Delivery systems are not the only parameter with room for improvement; optimizing payload chemical properties also holds significant potential. For instance, Biogen's intrathecal drug salanersen achieves annual dosing due to its highly efficient splice-switching properties, which are enabled by its unique ribosomal modification. Meanwhile, City Therapeutics is developing a novel siRNA design to generate more effective RNA interference trigger molecules.
Delivering conjugated oligonucleotides to extrahepatic tissues presents a multifaceted challenge requiring integrated approaches. Novartis plans to submit BLA applications for Avidity's AOC study therapies targeting DM1 and facioscapulohumeral muscular dystrophy in 2027 and 2028 respectively, with projected revenues exceeding billions of dollars from these products by 2030. Such success is poised to drive substantial investments across the biotechnology industry.
reference material :
Now with oligos: antibody-oligonucleotide conjugates are the new drug modality to watch. Nat Biotechnol. 2026 Jan;44(1):3-5.
Disclaimer: This article is intended solely for knowledge exchange, sharing, and popular science purposes, and does not involve commercial promotion. It should not be construed as medical guidance or medication recommendations. For copyright infringement inquiries, please contact us for removal.
Product recommendations for our company:
1.433212-75-0 https://www.bicbiotech.com/product_detail.php?id=6522
2.1170354-22-9 https://www.bicbiotech.com/product_detail.php?id=6523
3.20358-03-6 https://www.bicbiotech.com/product_detail.php?id=6524
4.81686-22-8 https://www.bicbiotech.com/product_detail.php?id=6525
5.61595-77-5 https://www.bicbiotech.com/product_detail.php?id=6526




