With the advancement of tumor biology research, human understanding of cell death regulatory mechanisms has continuously evolved, with their classification systems and molecular implications being progressively expanded and refined. From classical apoptosis (Apoptosis) to autophagy and ferroptosis, each newly discovered death mode has brought new hope for cancer therapy. In 2022, Tsvetkov et al. published a landmark study in Science, formally defining a novel copper-dependent programmed cell death mechanism—copperoptosis. This discovery not only filled the gap in metal ion-induced cell death mechanisms but also revealed a previously unknown profound connection between mitochondrial metabolism and cellular fate.

Figure 1. Article published in Science
Copper, as an essential trace element for life, serves as both a "catalyst" for cellular energy metabolism and a potentially lethal "poison" when present in excess. Tumor cells, due to their rapid proliferation and dependence on mitochondrial respiration, often exhibit unique copper metabolism characteristics. In recent years, with the rapid advancement of nanotechnology, scientists have ingeniously integrated the molecular mechanisms of copper-mediated apoptosis with nanodrug delivery systems, developing a series of novel therapies capable of precisely inducing copper-mediated apoptosis in tumor cells. These strategies not only overcome the drug resistance issues associated with conventional chemotherapy but also demonstrate remarkable synergistic potential when combined with immunotherapy, radiotherapy, and physical therapies.
Next, we will conduct an in-depth analysis of the molecular mechanisms underlying copper death, systematically review copper death-inducing strategies based on nanotechnology, and focus on exploring the latest research advancements in combination therapies. This aims to provide a scientific, objective, and comprehensive perspective on understanding this cutting-edge field, while envisioning its broad prospects for future precision oncology treatments.
I. Molecular Mechanism Analysis of Copper Death
Copper death is not merely copper poisoning, but rather a precisely regulated intracellular crisis. Its core mechanism involves abnormal accumulation of copper ions within cells, which directly attacks key components of the mitochondrial tricarboxylic acid cycle (TCA cycle), leading to protein toxic stress and cellular collapse.
(1) Fine balance of copper homeostasis
Under normal physiological conditions, cells maintain copper homeostasis through a sophisticated network. Copper ions primarily enter cells via the SLC31A1 (CTR1) transporter protein and are subsequently chelated by metallothionein (MT) or glutathione (GSH) to maintain extremely low free copper concentrations. Copper chaperone proteins (e.g., ATOX1, COX17) are responsible for directing copper delivery to specific organelles (e.g., Golgi apparatus, mitochondria) and integrating it into functional proteins (e.g., SOD1, cytochrome c oxidase). When copper overload occurs, the ATP7A/B transporter protein pumps excess copper out of the cell. However, once this balance is disrupted—particularly when copper concentration becomes uncontrolled within mitochondria—copper death is initiated.
(2) Core pathway: Acylated protein aggregation and iron-sulfur cluster depletion
The uniqueness of copper sequestration lies in its target mechanism. Studies have revealed that after copper ions are transported into cells by carriers such as Elesclomol, the key regulatory factor FDX1 (ferritin 1) exerts a dual role:
Reduction effect: FDX1 reduces the less toxic Cu²⁺ to the highly toxic Cu⁺.
Binding induction: Cu⁺ directly binds to enzymes undergoing lipooylation modification in the TCA cycle (primarily DLAT and DLST). This binding leads to abnormal oligomeric aggregation of acylated proteins, disrupting the normal mitochondrial structure.
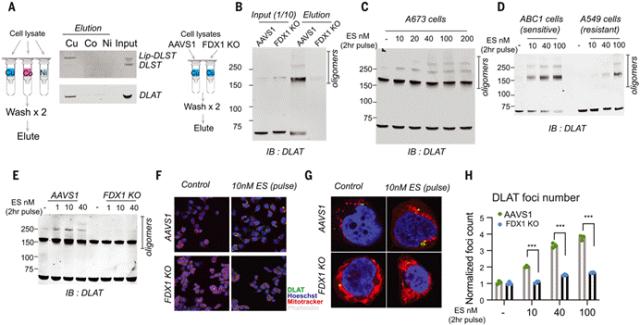
Figure 2. Copper directly binds and promotes the oligomerization of acylated DLAT
Loss of iron-sulfur clusters: Concurrently, copper overload leads to reduced protein stability and degradation of iron-containing sulfur clusters (Fe-S clusters). Fe-S clusters are critical coenzymes for mitochondrial electron transport and enzyme activity, and their loss further exacerbates mitochondrial dysfunction.
The aforementioned process induces intense proteotoxic stress, leading to upregulation of heat shock proteins (e.g., Hsp70) and ultimately resulting in cell death. Notably, copper-induced apoptosis is highly independent of inhibitors of typical death pathways such as BAX/BAK (apoptosis), GPX4 (ferroptosis), or Caspases (pyroptosis).
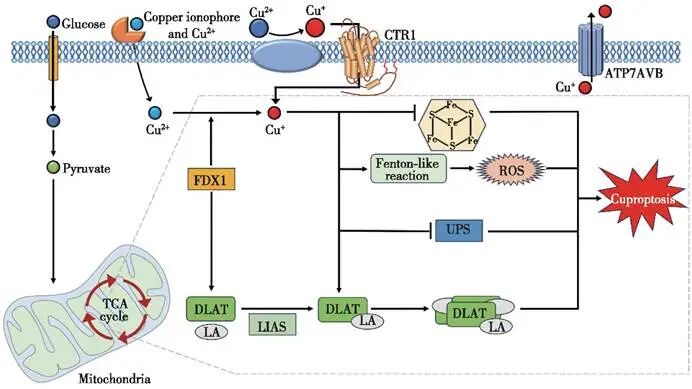
Figure 3. Molecular mechanism of copper death schematic diagram
II. Copper Death Nanomedicine
Although the mechanism of copper-mediated cell death is well understood, direct utilization of copper ions for therapeutic purposes faces significant challenges: copper ions are readily cleared from the bloodstream through protein binding and lack tumor targeting specificity, which may lead to toxicity in normal tissues (e.g., liver, nerves). The emergence of nanotechnology offers a perfect solution to address this issue.
(1) Multiple advantages of nanocarriers
Copper-based nanomedicines or nanosystems loaded with copper ion carriers possess the following core advantages:
Passive targeting (EPR effect): Leveraging the high permeability and retention effect of tumor blood vessels, nanoparticles can accumulate at the tumor site.
Microenvironment Response: Designing pH-, GSH-, or enzyme-responsive nanomaterials to achieve targeted release of copper ions within tumor cells.
Overcoming drug resistance: Nanocarriers can bypass efflux pumps such as P-gp, thereby increasing intracellular drug concentration.
(2) Diversification of induction mechanisms
Current nanotechnology strategies primarily include:
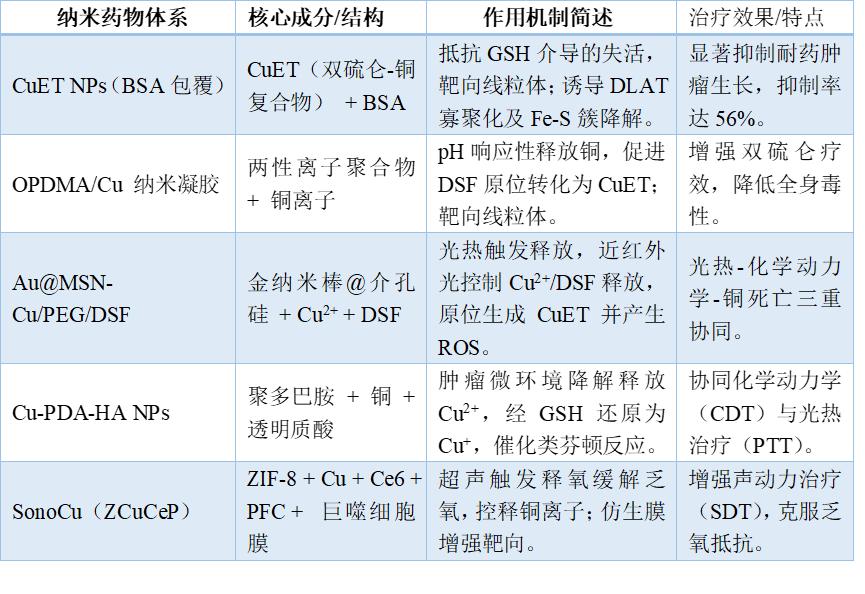
In situ synthesis of toxic complexes: For example, co-delivery of disulfiram (DSF) and copper ions generates highly toxic CuET complexes intracellularly in tumor cells, directly inducing copper-mediated apoptosis and inhibiting proteasome activity.
Interference with copper efflux: Utilizing nanoscale RNA interference technology to downregulate ATP7A/B, thereby blocking copper efflux and artificially inducing intracellular copper overload.
Antioxidant depletion: Certain nanomaterials can simultaneously deplete GSH, thereby relieving its chelating effect on copper ions and "unlocking" the toxicity of copper.
Table 1. Representative copper death-inducing nanomedicines and their mechanisms of action
III. Combination Therapy for Copper Death
Copper-deficient nanomedicines do not exist in isolation; their true efficacy lies in the "synergistic interaction" with other therapeutic modalities. Through multi-mechanism collaboration, a therapeutic effect greater than the sum of individual components ("1+1>2") can be achieved.
(1) Copper-induced cell death + immunotherapy: Activation of antitumor immunity
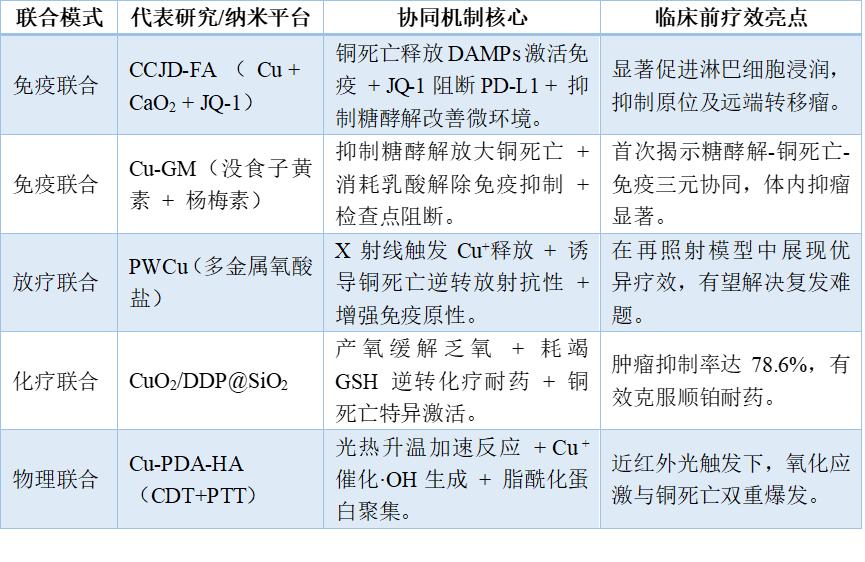
Copper-induced death often exhibits characteristics of immunogenic cell death (ICD). Dying tumor cells release damage-associated molecular patterns (DAMPs), such as calreticulin (CRT), HMGB1, and ATP, which can activate dendritic cells and subsequently recruit CD8⁺ T cells to kill tumors.
Reversal of immunosuppression: Studies have found that copper death can reduce lactate levels in the tumor microenvironment (by inhibiting glycolysis), thereby alleviating the inhibitory effects of lactate on immune cells.
Combined checkpoint inhibitors: Although copper overload may upregulate PD-L1, the combination of PD-1/PD-L1 antibodies or BRD4 inhibitors (e.g., JQ-1) can effectively block immune evasion and significantly enhance tumor suppression effects at distant sites ("bystander effect").
(2) Copper-induced necrosis + radiotherapy/chemotherapy: dual impact and sensitization
Radiation sensitization: ROS generated by radiotherapy can exacerbate copper-induced mitochondrial damage. Certain copper-containing nanomaterials (such as polymetallooxides) can precisely release Cu⁺ under X-ray irradiation, not only directly inducing copper death but also reversing radiation resistance by disrupting DNA repair mechanisms.
Overcoming chemotherapy resistance: The copper-mediated death mechanism operates independently of traditional apoptosis pathways, thus remaining effective against cells resistant to cisplatin, paclitaxel, and other agents. Additionally, copper ion carriers can inhibit the P-gp efflux pump, enhancing the intracellular accumulation of chemotherapeutic agents. For instance, DOX/Cu complexes exert a "synthetic lethality" effect by simultaneously damaging DNA and mitochondria, significantly inhibiting hepatocellular carcinoma.
(3) Copper-induced cell death + physical therapy (light/sonic/chemodynamic)
Chemodynamic kinetics (CDT): Copper ions themselves serve as excellent Fenton-like reaction catalysts. The Cu⁺ released by nanomedicines can convert excess H2O2 within tumors into highly toxic ·OH radicals, triggering an oxidative storm that forms a positive feedback loop with the protein toxicity stress induced by copper-induced cell death.
Photothermal/Photodynamic: The photothermal effect can accelerate the release of copper ions and metabolic reaction rates; the singlet oxygen generated by photodynamic therapy can consume GSH, further releasing copper and enhancing copper death efficiency.
Table 2. Combined Therapy Strategies and Synergistic Mechanisms for Copper Necrosis
IV. Conclusion
The discovery of copper death has opened up new research paradigms and therapeutic strategies in the field of tumor treatment. It represents not only a novel form of cell death but also a pivotal hub connecting metal metabolism, mitochondrial function, and cellular fate. Nanotechnology-based delivery strategies can overcome the non-specific distribution limitations of copper ions, enabling tumor microenvironment-responsive release and effectively decoupling the therapeutic effects of copper from systemic toxic side effects. From single copper ion delivery to deep synergies with immunotherapy, chemoradiotherapy, and physical therapies, copper death nanomedicines demonstrate exciting therapeutic prospects. Particularly, their unique advantages in overcoming tumor resistance, reversing immunosuppressive microenvironments, and inducing distant antitumor effects provide novel insights for addressing current clinical treatment bottlenecks.
However, the path to clinical application remains challenging. Specific biomarkers for copper death require further validation, while long-term biosafety of nanomedicines, in vivo metabolic fate, and standardization of large-scale production pose urgent challenges. Additionally, the differential sensitivity of various tumor types to copper death and underlying molecular determinants necessitate deeper elucidation. Looking ahead, with the application of cutting-edge technologies such as single-cell sequencing and spatial metabolomics, coupled with comprehensive decoding of copper homeostasis regulatory networks, precision therapy strategies based on copper death will become more mature. Future therapeutic approaches induced by copper death are expected to be integrated into comprehensive cancer treatment regimens, serving as a critical component for improving patient outcomes. As the regulatory mechanisms of copper metabolism are further elucidated and applied, this field is driving paradigm shifts in antitumor therapies and holds promise for reshaping the landscape of precision oncology.
References :
1. Tsvetkov P, Coy S, Petrova B, Dreishpoon M, Verma A, Abdusamad M, Rossen J, Joesch-Cohen L, Humeidi R, Spangler RD, Eaton JK, Frenkel E, Kocak M, Corsello SM, Lutsenko S, Kanarek N, Santagata S, Golub TR. Copper induces cell death by targeting lipoylated TCA cycle proteins. Science. 2022 Mar 18;375(6586):1254-1261. doi: 10.1126/science.abf0529. Epub 2022 Mar 17. Erratum in: Science. 2022 Apr 22;376(6591):eabq4855. doi: 10.1126/science.abq4855. PMID: 35298263; PMCID: PMC9273333.
2. Chan N, Willis A, Kornhauser N, Ward MM, Lee SB, Nackos E, Seo BR, Chuang E, Cigler T, Moore A, Donovan D, Vallee Cobham M, Fitzpatrick V, Schneider S, Wiener A, Guillaume-Abraham J, Aljom E, Zelkowitz R, Warren JD, Lane ME, Fischbach C, Mittal V, Vahdat L. Influencing the Tumor Microenvironment: A Phase II Study of Copper Depletion Using Tetrathiomolybdate in Patients with Breast Cancer at High Risk for Recurrence and in Preclinical Models of Lung Metastases. Clin Cancer Res. 2017 Feb 1;23(3):666-676. doi: 10.1158/1078-0432.CCR-16-1326. Epub 2016 Oct 21. Erratum in: Clin Cancer Res. 2020 Sep 15;26(18):5051. doi: 10.1158/1078-0432.CCR-20-3177. PMID: 27769988.
3. Jin J, Ma M, Shi S, Wang J, Xiao P, Yu HF, Zhang C, Guo Q, Yu Z, Lou Z, Teng CB. Copper enhances genotoxic drug resistance via ATOX1 activated DNA damage repair. Cancer Lett. 2022 Jun 28;536:215651. doi: 10.1016/j.canlet.2022.215651. Epub 2022 Mar 18. PMID: 35315340.
4. Voli F, Valli E, Lerra L, Kimpton K, Saletta F, Giorgi FM, Mercatelli D, Rouaen JRC, Shen S, Murray JE, Ahmed-Cox A, Cirillo G, Mayoh C, Beavis PA, Haber M, Trapani JA, Kavallaris M, Vittorio O. Intratumoral Copper Modulates PD-L1 Expression and Influences Tumor Immune Evasion. Cancer Res. 2020 Oct 1;80(19):4129-4144. doi: 10.1158/0008-5472.CAN-20-0471. Epub 2020 Aug 18. PMID: 32816860.
5. Ge EJ, Bush AI, Casini A, Cobine PA, Cross JR, DeNicola GM, Dou QP, Franz KJ, Gohil VM, Gupta S, Kaler SG, Lutsenko S, Mittal V, Petris MJ, Polishchuk R, Ralle M, Schilsky ML, Tonks NK, Vahdat LT, Van Aelst L, Xi D, Yuan P, Brady DC, Chang CJ. Connecting copper and cancer: from transition metal signalling to metalloplasia. Nat Rev Cancer. 2022 Feb;22(2):102-113. doi: 10.1038/s41568-021-00417-2. Epub 2021 Nov 11. PMID: 34764459; PMCID: PMC8810673.
6. Bost M, Houdart S, Oberli M, Kalonji E, Huneau JF, Margaritis I. Dietary copper and human health: Current evidence and unresolved issues. J Trace Elem Med Biol. 2016 May;35:107-15. doi: 10.1016/j.jtemb.2016.02.006. Epub 2016 Mar 5. PMID: 27049134.
Author: Li Ya, primarily engaged in drug quality control and analytical testing.
Disclaimer: This article is intended solely for knowledge exchange, sharing, and popular science purposes, and does not involve commercial promotion. It should not be construed as medical guidance or medication recommendations. For copyright infringement inquiries, please contact us for removal.
Product recommendations for our company:
1.383137-88-0 https://www.bicbiotech.com/product_detail.php?id=6532
2.216387-73-4 https://www.bicbiotech.com/product_detail.php?id=6533
3.36877-69-7 https://www.bicbiotech.com/product_detail.php?id=6534
4.1315568-19-4 https://www.bicbiotech.com/product_detail.php?id=6535
5.53947-84-5 https://www.bicbiotech.com/product_detail.php?id=6536




