Since the 19th century, infectious diseases have remained a critical public health challenge threatening human health and social stability. Their occurrence and prevalence are driven not only by pathogen mutations but also closely linked to accelerated global population mobility, disparities in public health conditions, and advancements in medical technology. This article focuses on reviewing progress in drug development for tuberculosis, analyzing the clinical value and evolutionary trends of various mechanisms of action and innovative strategies. By examining the overall landscape of anti-infective drug development in China, it summarizes the phased achievements and major challenges of domestic innovation, aiming to provide insights for comprehensively understanding the global landscape and China's approach to infectious disease drug development.
Tuberculosis (TB) is a chronic infectious disease caused by infection with Mycobacterium tuberculosis, which is widely prevalent worldwide. Pulmonary tuberculosis (PTB) represents the most common form of TB. The causative pathogens of TB belong to the Mycobacterium tuberculosis complex, including Mycobacterium tuberculosis, Mycobacterium bovis, Mycobacterium africanum, and Mycobacterium avium. Mycobacterium tuberculosis accounts for 90% of human pulmonary tuberculosis cases. According to the World Health Organization (WHO) Global TB Report 2025, there were 10.7 million new TB cases globally in 2024, with an incidence rate of 131 per 100,000 population; TB caused over 1.2 million deaths, once again making it the leading cause of death among infectious diseases. The TB epidemic situation in China remains concerning. Although both the incidence and mortality rates of TB have shown a downward trend, there is still a significant gap to achieving the goal of ending TB.
marketed drug
Antituberculosis drugs have been clinically used for over half a century. For patients with first-line-sensitive pulmonary tuberculosis, the cure rate after six months of standardized treatment with first-line antituberculosis drugs can reach 85%. However, due to drug resistance in patients with drug-resistant tuberculosis to most first-line antituberculosis drugs, the overall treatment success rate has dropped below 60%. Traditional antituberculosis drugs are limited by their prolonged treatment courses, severe adverse reactions, and increasingly severe drug resistance, making the development of novel, highly effective treatment regimens urgently necessary. Currently available new antituberculosis drugs include bedaquiline (first approved in the United States in 2012 and in China in 2016), delamanid (first approved in the European Union in 2014 and in China in 2018), and putamanid (first approved in the United States in 2019 and in China in 2024). However, due to their high cost and the observed resistance to bedaquiline, the development of new antituberculosis drugs is particularly crucial.
Bedaquiline belongs to the diarylquinoline class of drugs. In December 2012, the U.S. FDA expedited its approval for the treatment of multidrug-resistant tuberculosis (MDRTB) due to its demonstrated efficacy and safety. Bedaquiline exhibits potent bactericidal activity against multidrug-resistant Mycobacterium tuberculosis both in vivo and in vitro. Its mechanism of action primarily involves inhibition of mycobacterial adenosine triphosphate (ATP) synthase. ATP synthase is a key enzyme located in the inner mitochondrial membrane of Mycobacterium tuberculosis, responsible for generating energy to support metabolic processes such as synthesis and degradation in growing Mycobacterium tuberculosis cells. Bedaquiline binds to the oligo/protein lipid subunit c or δ of the ATP synthase F region, thereby disrupting the activity of the ATP synthase proton pump and inhibiting ATP synthesis. This prevents Mycobacterium tuberculosis from obtaining ATP for energy production, achieving its bactericidal effect. However, bedaquiline is insensitive to human ATP synthase, ensuring minimal toxicity targeting human ATP synthase and negligible potential for interaction with it.
However, the issue of bedaquiline resistance cannot be overlooked, with a baseline phenotypic resistance rate of 2.4% among multidrug-resistant patients. To reduce the incidence of resistance, the study design protocol is critical. In a retrospective observational study, Barilar et al. performed whole-genome sequencing on 704 rifampin-resistant Mycobacterium tuberculosis strains from Mozambique. Sixty-one strains (9%) exhibited bedaquiline resistance, with the bedaquiline resistance rate rising from 3% in 2016 to 14% in 2021. Additionally, certain newer anti-tuberculosis drugs prolong the QT interval, increasing the risk of arrhythmias and sudden cardiac death. Jin et al. employed an observational prospective cohort study and found that in patients treated with bedaquiline or delamanide, the mean maximum increase in heart rate-corrected QT interval (QTc) over the first 6 months was 37.5 ms (IQR: 17.8–56.8 ms). During treatment, regimens involving bedaquiline and delamanide showed similar QTc prolongations. The increase in the number of anti-tuberculosis drugs was associated with QTc prolongation, but values returned to baseline after discontinuation.
Dramani is a nitro-dihydroimidazole derivative that primarily disrupts cell wall synthesis by inhibiting the production of methoxymycobenic acid and ketomycobenic acid, components of the Mycobacterium tuberculosis (MTB) cell wall. This mechanism facilitates drug penetration into MTB cells to exert bactericidal effects, which are effective against both replicating, dormant, and intracellular MTB. Additionally, studies have demonstrated that dramani inhibits aerobic respiration in MTB, suggesting the drug may induce respiratory failure by suppressing MTB aerobic respiration, thereby achieving its bactericidal action. Following oral administration of dramani for 4–8 hours, peak plasma concentrations are achieved, enabling rapid demonstration of potent in vitro bactericidal activity with strong inhibitory effects against MTB.
In a prospective multicenter study, 608 patients with multidrug-resistant/rifampin-resistant tuberculosis (MDR/RR-TB) were enrolled. During the 24-week dramanid treatment period, a total of 710 adverse reactions were reported in 325 patients (53.5%). The top six most frequent adverse reactions were hematologic impairment (143 cases, 23.5%), QT interval prolongation on electrocardiogram (ECG) (114 cases, 18.8%), hepatotoxicity (85 cases, 14.0%), gastrointestinal reactions (41 cases, 6.7%), peripheral neuropathy (25 cases, 4.1%), and psychiatric disorders (21 cases, 3.5%). QT interval prolongation on ECG predominantly occurred at week 12, with 21 cases (3.5%) reporting severe adverse events; psychiatric disorders were observed in 7 patients (1.2%), including 2 cases (0.3%) with severe psychiatric disturbances. The overall safety profile of the dramanid regimen was favorable; however, the incidence of QT interval prolongation in the Chinese patient population was higher than reported in international studies.
Pertomanid is an oral nitroimidazole-class antibacterial agent developed under license from Novartis by the TB Alliance as part of the BPaL (Betaquinoline, Pertomanid, and Linzimidazole) regimen for the treatment of drug-resistant tuberculosis. Initially approved in the United States in 2019, pertomanid has since been rolled out globally, including in the European Union, Africa, the Americas, and Asia, with approval now available in over 60 countries and regions. Ongoing post-marketing studies have validated the efficacy and safety of the BPaL regimen. The 2022 WHO Guidelines on Drug Resistance explicitly recommend the BPaLM/BPaL regimen (gamoxacin in the treatment of fluoroquinolone-sensitive patients) for multidrug-resistant or rifampin-resistant tuberculosis, with the combination regimen containing betaquinoline, pertomanid, and linzimidazole being the sole six-month treatment regimen endorsed by the guidelines. As the latest globally marketed anti-tuberculosis drug, pertomanid exhibits multiple mechanisms of action: it inhibits mycotoxin biosynthesis and disrupts cell wall synthesis to eradicate actively replicating bacteria under aerobic conditions; it also suppresses non-replicating bacteria under anaerobic conditions by inducing respiratory toxicity via nitric oxide release and reducing intracellular adenosine triphosphate (ATP) levels. Recent studies demonstrate that pertomanid targets the pentose phosphate pathway, leading to pentose phosphate accumulation and subsequent toxic accumulation of methylaldehyde, thereby inducing cell growth arrest. This study confirms that Puthomanib exhibits a mechanism of action entirely distinct from other drugs used for tuberculosis treatment. Its multi-faceted mechanism of action endows Puthomanib with potent bactericidal and sterilizing capabilities, thereby ensuring sufficiently high efficacy with a shortened treatment duration. In completed Phase II/III clinical trials, Puthomanib demonstrated favorable efficacy and safety profiles. Key completed clinical trials include NiX-TB, ZeNiX, SimpliciTB, and TB-PRACTECAL, among others. These trials also serve as evidence supporting the WHO guidelines recommending the BPaLM/BPaL regimen.
Drug under development
In recent years, driven by the growing demand for treatment of drug-resistant tuberculosis, global research and development of novel anti-tuberculosis drugs has intensified significantly, with several candidate drugs featuring novel mechanisms of action achieving positive progress in clinical trials (Table 1). Overall, among the currently investigational anti-tuberculosis drugs, desopentadienyl phosphate-β-D-ribose 2'-epimerase (DprE1) inhibitors and novel oxazolidinone-based protein synthesis inhibitors exhibit the highest activity. Additionally, several small-molecule drugs targeting novel mechanisms or demonstrating synergistic sensitization effects are progressively advancing into clinical research stages.
Table 1: Selected investigational anti-tuberculosis drugs
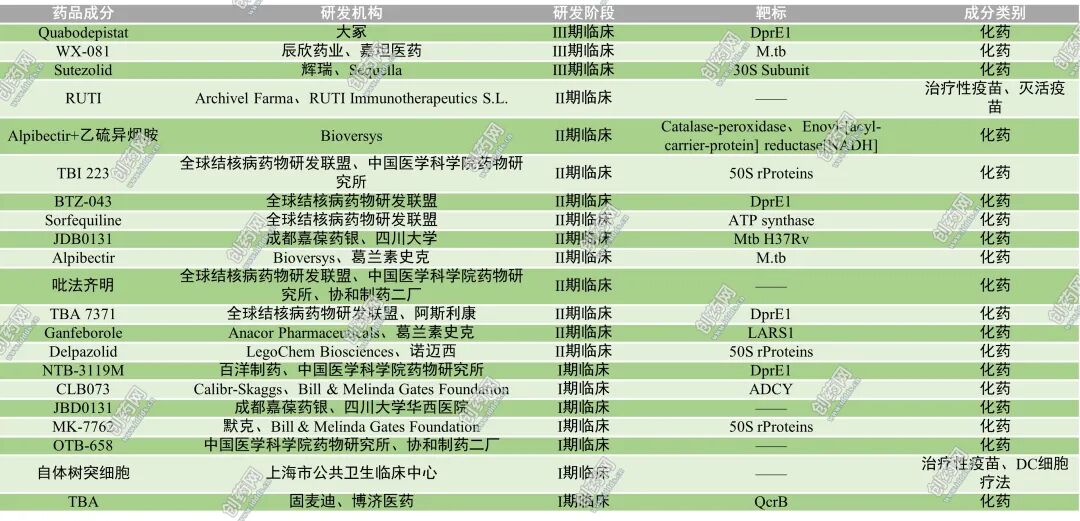
First, DprE1 inhibitors have emerged as one of the most promising classes of antituberculosis drugs in recent years. DprE1 is a key enzyme in the arabinopolysaccharide biosynthesis pathway of the Mycobacterium tuberculosis cell wall; inhibition of this enzyme blocks arabinopolysaccharide production, thereby disrupting cell wall formation and leading to bacterial death. Consequently, these drugs exhibit significant bactericidal activity against both drug-sensitive and drug-resistant Mycobacterium tuberculosis strains. Currently, representative drugs with rapid clinical progress include Quabodepistat and BTZ-043. In April 2025, The Lancet Infectious Diseases published a randomized controlled clinical trial evaluating the efficacy and safety of Quabodepistat combined with bedaquiline and delamarnine in treating adult pulmonary tuberculosis. This study established the first fully novel-drug-based antituberculosis combination regimen in nearly 50 years, demonstrating favorable safety and tolerability profiles along with potent early bactericidal activity, indicating potential for further large-scale clinical development. Additionally, Phase Ib/IIa clinical trial results published in The Lancet Microbe in February 2025 demonstrated that BTZ-043 exhibited favorable safety, pharmacokinetic characteristics, and antibacterial activity in tuberculosis patients, further validating the significant potential of DprE1 inhibitors as novel antituberculosis agents.
Secondly, novel oxazolidinone-based protein synthesis inhibitors represent another category of rapidly advancing anti-tuberculosis candidate drugs. These compounds enhance the stability of microsomes and hepatocytes, exhibit no inhibitory effect on the CYP enzyme system, demonstrate high oral bioavailability, and exhibit antibacterial activity equal to or superior to linezolid against both sensitive and drug-resistant Mycobacterium tuberculosis strains, with better safety profiles compared to linezolid. Representative drugs currently in clinical development include Delpazolid, Sutezolid, TBI-223, and MK-7762. In July 2025, The Lancet Infectious Diseases published two Phase IIb clinical trials evaluating the efficacy of Delpazolid and Sutezolid in tuberculosis treatment. The results demonstrated that both drugs exhibited favorable antibacterial efficacy and safety when combined with the regimen comprising betacarbazone, delamanid, and moxifloxacin. These novel oxazolidinone compounds hold promise for mitigating or replacing the toxicity associated with long-term linezolid use and may become a critical component in the treatment of multidrug-resistant tuberculosis, complementing or substituting existing therapeutic regimens (such as the WHO-recommended BPaLM regimen).
In addition to the aforementioned two major therapeutic mechanisms, several novel targeted anti-tuberculosis drugs have gradually entered clinical research stages. For example, Alpibectir is a small-molecule drug targeting mycobacterial transcriptional regulators, acting on VirS to enhance the biological activity of ethionamide both in vitro and in vivo. By stimulating the unconventional MymA pathway, Alpibectir overcomes drug resistance induced by EthA mutations. In a Phase I, double-blind, randomized, placebo-controlled study, Alpibectir was administered orally to human subjects for the first time. Results demonstrated generally favorable tolerability, with no serious adverse events reported among participants in single-dose escalation or multiple-dose escalation trials.
Furthermore, Ganfeborole represents a novel class of benzoxaborazole-based antituberculosis drugs and is the first identified inhibitor of Mycobacterium tuberculosis leucine-tRNA synthetase. A Phase IIa trial evaluated the efficacy and safety of Ganfeborole in rifampin-sensitive, first-line-treated pulmonary tuberculosis patients, demonstrating its bactericidal activity against Mycobacterium tuberculosis and acceptable safety profile, making it a potential candidate for combination therapy in pulmonary tuberculosis.
Overall, current tuberculosis drug development is progressively shifting from traditional antibacterial mechanisms toward novel targets and novel mechanisms of action, while optimizing therapeutic efficacy through multi-drug combination strategies. With the ongoing advancement of clinical trials for these candidate drugs, it is anticipated that the treatment duration for tuberculosis will be further shortened, adverse effects minimized, and therapeutic outcomes for patients with drug-resistant tuberculosis improved.
【 reference material 】
1. Fuchs SP, Mondragon PG, Zabizhin R, et al. Transient rapamycin treatment avoids unwanted host immune responses toward AAV-delivered anti-HIV antibodies. Nat Commun, 2025, 16(1): 8906.
2. Wallis ZK, Wei Y, Ceraso LM, et al. Novel perineural pathways and the dynamics of SIV-infected macrophage trafficking out of the central nervous system. Am J Pathol, 2025, 195(11): 2233-2246.
3. Zhu Biao, He Lingling. Recent Advances in Antiretroviral Drugs for Human Immunodeficiency Virus. Zhejiang Medical Journal, 2025,47(9):897-902.
4. Xia Yu, Sun Yanying, Kang Dongwei, et al. Advances in AIDS drug research based on target structures. China Journal of Medicinal Chemistry, 2025,35(1):39-58.
5. Shu Fuyan, Yang Ming. Research progress on bedaquiline for the treatment of multidrug-resistant tuberculosis. Journal of Clinical Pulmonology, 2023,28(2):286-290.
6. Abulimiti Abdulkadir, Li Peibo, Xie Jianping. Global Advances in Tuberculosis Drug Development in 2024: Multi-target Strategies, Breakthroughs in Drug Resistance Mechanisms, and Personalized Therapy. Journal of Tuberculosis and Lung Diseases, 2025,6(5):587-597.
7. Ren Zhinan, Peng Xuqing, Wang Jie, et al. The ultimate weapon: rapid cure with Puthomanib accelerates the achievement of the "end-of-tuberculosis" goal. Proceedings of the 35th China Anti-Tuberculosis Association National Academic Conference and the 4th China Anti-Tuberculosis Science and Technology Awards Ceremony (Clinical Volume). Shenyang Hongqi Pharmaceutical Co., Ltd., 2024:161–162.
8. Dingxiangyuan Insight database; retrieval date: March 10,2026.
Disclaimer: This article is intended solely for knowledge exchange, sharing, and educational purposes only. It does not constitute commercial promotion and should not be regarded as medical guidance or medication recommendations. In case of any copyright infringement, please contact us for removal.
Our product recommendations:
1.13558-31-1 https://www.bicbiotech.com/product_detail.php?id=6537
2.21018-71-3 https://www.bicbiotech.com/product_detail.php?id=6538
3.960373-35-7 https://www.bicbiotech.com/product_detail.php?id=6539
4.1418610-53-3 https://www.bicbiotech.com/product_detail.php?id=6540
5.21658-70-8 https://www.bicbiotech.com/product_detail.php?id=6541




