"Knowing full well this is an accident, would you still come?"
In the long journey of innovative drug development, the "Valley of Death" is a term that strikes fear into the hearts of all practitioners. It is not a specific geographical location but a vivid metaphor—describing the chasm between fundamental research breakthroughs and the successful commercialization of drugs, a gap that leads to the failure of the vast majority of drug candidates.
I. The Origin and Conceptual Evolution of the "Valley of Death"
The concept of the "Valley of Death" dates back to 1991, when Mohawk Research Corporation first employed it in a report to the U.S. Department of Energy's Argonne National Laboratory to describe the funding gap between product prototyping and commercialization. In 1998, the U.S. House Science Committee reiterated this metaphor in its report "Unlocking Our Future: Toward a New National Science Policy," highlighting the funding shortfall between basic research and product development. This concept accurately captures the structural fracture in the technological innovation chain—a "no man's land" between government-funded basic research and the commercialization driven by industry capital, where numerous promising discoveries are abandoned due to lack of sustained support.
In the field of biomedicine, the connotation of the "Valley of Death" has been further deepened and specified. In 2008, Declan Butler, a senior journalist at Nature, published a signed article titled "Crossing the 'Valley of Death'," highlighting the increasingly widening gap between biomedical researchers and patients. He called on the National Institutes of Health (NIH) to make adjustments in terms of conceptual frameworks, policy directions, and financial support to facilitate translational research from the laboratory to clinical settings. A subsequent academic study published that same year provided even more incisive insights: approximately 90% of drug candidates fail to demonstrate therapeutic efficacy in human subjects during the transition from preclinical to clinical trials.
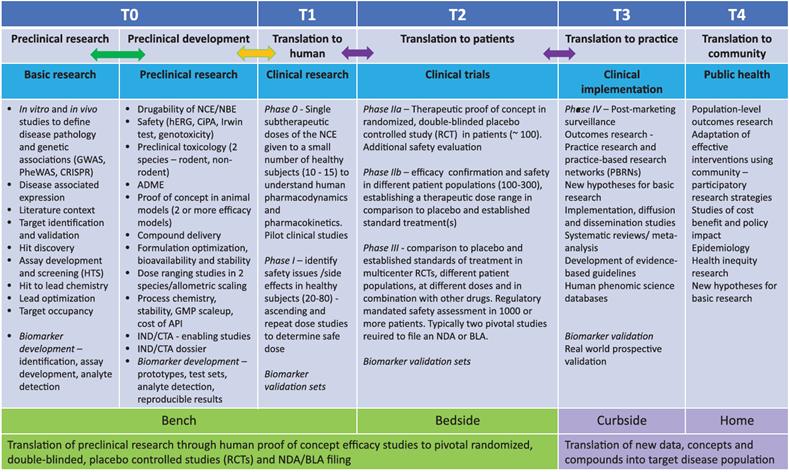
Figure 1: Bidirectional Transformation Framework – Stages and Key Transition Nodes. T0–T4 represent the distinct transformation stages in the drug discovery and development process. The bidirectional arrows illustrate the feedback mechanisms at each stage, enabling re-evaluation, optimization, and prioritization of various aspects of the transformation process based on actual outcomes. The columns under the headings in the blue bar describe the specific activities involved in each transformation stage. The green and purple bars, respectively, summarize the key tasks across stages T0–T4 from the perspective of the general concept of "from laboratory to clinical (and beyond)."
The contemporary academic community has provided a more refined definition of the "Valley of Death." According to a perspective published in Current Protocols in 2021, translational failures in drug development actually encompass two levels: T1 translational failure, which refers to the inability to translate basic research findings into clinical candidates (i.e., failure during the transition from preclinical to Phase I clinical trials); and T2 translational failure, which denotes the failure to sequentially demonstrate the efficacy and safety of a drug in Phase II and Phase III clinical trials. These two levels collectively constitute the "Valley of Death" in the field of drug development.
II. The Quantitative Portrait of the "Valley of Death"
2.1 A staggering failure rate
Drug discovery and development is a lengthy, costly, and high-risk process. On average, it takes 10–15 years for a new drug to progress from research and development to clinical approval, with costs exceeding $1–2 billion. For any pharmaceutical company or academic institution, the advancement of a drug candidate through rigorous preclinical optimization to Phase I clinical trials represents a significant achievement. However, among drug candidates that enter clinical trials, nine out of ten fail during Phase I, II, or III clinical trials or regulatory approval stages. It is important to note that this 90% failure rate applies solely to drug candidates that reach Phase I clinical trials and does not include those in the preclinical stage. When the preclinical phase is included, the failure rate in drug discovery/development exceeds 90%.
2.2 In-depth Analysis of Failure Causes
What exactly led to such a devastating failure rate? A systematic analysis of clinical trial data from 2010 to 2017 identifies four major categories of contributing factors:
The lack of clinical efficacy (40%-50%) is the primary cause of failure. This reflects the "original sin" in the basic research phase—the limited predictive power of animal models. A compound that demonstrates remarkable efficacy in transgenic mouse models may prove entirely ineffective when administered to humans. This gap between animal and human models constitutes the core reason for T1 translation failures.
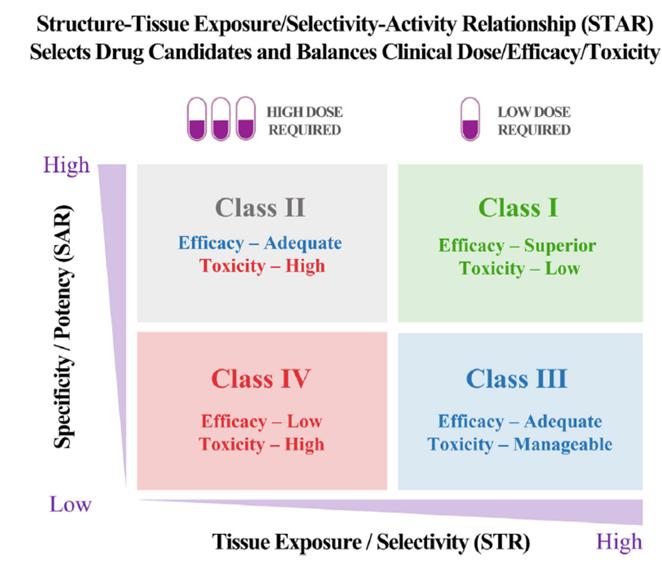
Figure 2 illustrates the Structure-Organization Selectivity/Exposure Activity Relationship (STAR), which enables the screening of superior drug candidates and balances clinical dosage, efficacy, and toxicity, thereby optimizing drugs to facilitate successful clinical drug development.
Uncontrolled toxicity (30%) ranks second. Such failures typically occur during late clinical stages, when drugs are tested in broader patient populations, and previously undetected toxicities are suddenly revealed. Certain toxicity mechanisms cannot be effectively predicted in animal models, such as certain immune-related adverse reactions.
Poor drug-like properties (10%-15%) were the primary cause of failures in the 1990s, accounting for 30%-40% of failed cases. This proportion has now significantly declined, thanks to the industry's emphasis on early optimization of ADME (absorption, distribution, metabolism, excretion) characteristics. Drug-like properties such as solubility, permeability, and metabolic stability have become core criteria for candidate compound screening.
Commercial factors and strategic planning (10%) should not be overlooked either. Changes in therapeutic priorities, pipeline overlaps resulting from corporate mergers and acquisitions, and shifts in market dynamics may abruptly halt a project that was technically promising.
III. The Multidimensional Causes of the Valley of Death
3.1 Scientific Level: The Fundamental Limitations of Predictive Power
From a scientific perspective, the existence of the "Valley of Death" is rooted in the inherent complexity of biology itself. The species differences between animal models and humans constitute the primary reason for the failure of T1 translation. As emphasized in the 2024 American Society of Toxicology continuing education course, "insufficient understanding of the essence of the translation process" and "failure to effectively integrate data from different pharmacologically relevant species" are key factors contributing to these failures.
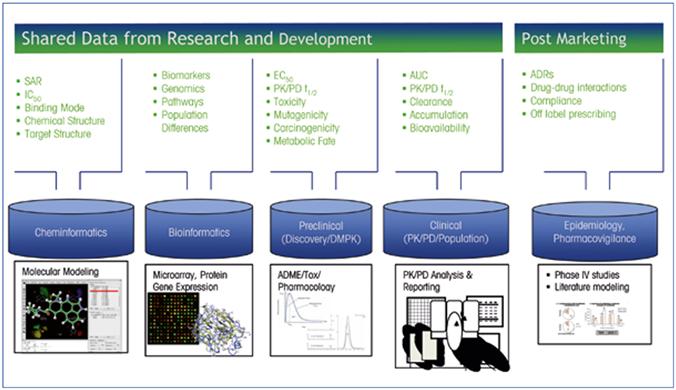
Figure 3: Example of data to be shared for optimizing translational science
The deeper issue lies in the fact that our understanding of disease mechanisms remains at an elementary stage. Even drugs with highly well-defined target mechanisms may fail in large-scale clinical trials. Although the target mechanism of the PCSK9 antibody Repatha is highly reliable, it did not achieve the expected therapeutic efficacy in the Fourier trial involving 27,000 participants. This case serves as a warning: biology is far more complex than we imagine.
3.2 Financial Dimension: The Mismatch Between Capital Supply and Demand
The concept of the "Valley of Death" was first proposed in the 1990s, with its core focus precisely on the funding gap. This issue is particularly acute in the biopharmaceutical sector. The development of innovative drugs is characterized by "high risk, high investment, and long cycles." On average, it takes 10–15 years from target discovery to final market approval, with R&D costs reaching $1–2 billion.
Funding misallocation manifests at two levels. First, the temporal mismatch between risk and return: early-stage R&D carries the highest risks but requires relatively little funding; later stages involve reduced risks but demand a dramatic increase in funding. Second, a structural gap in capital supply: high-risk early-stage projects struggle to attract commercial capital, while government funding favors basic research, creating a funding "no man's land."
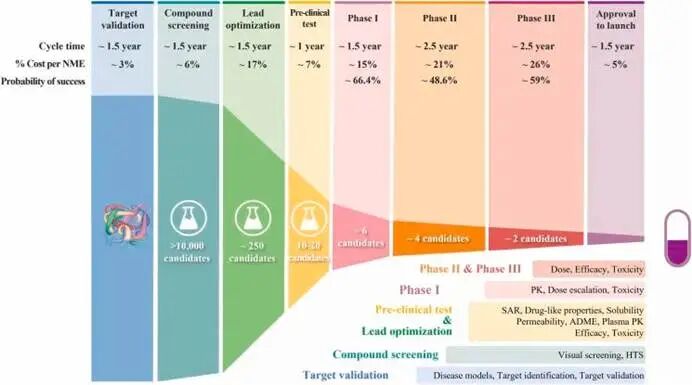
Figure 4. The process of drug discovery and development and the cost at each step
3.3 Organizational Level: Disciplinary Barriers and Decision-Making Dilemmas
The development of innovative drugs requires close collaboration across multiple disciplines, including chemistry, biology, medicine, pharmacy, toxicology, and statistics. However, practical implementation often faces communication barriers and conflicts of interest between these disciplines. Inadequate decision-making mechanisms also contribute to the "Valley of Death." Regarding early-stage data, the industry adopts two distinctly different strategic approaches: one advocates for "seeing the big picture" (pick the winners), asserting that any project's success requires enduring numerous challenges and should not be easily discouraged by failure signals; the other promotes "eliminating underperformers" (kill the losers), arguing that given the difficulty in assessing success potential at early stages, project diversity should be maximized, with projects terminated decisively once data indicate limited prospects. Both strategies have their merits, but both necessitate precise balancing between scientific uncertainty, commercial opportunities, and resource constraints.
3.4 Regulatory Level: The Challenge of a Lagging Framework
Changes in regulatory policies and uncertainties in approval requirements constitute macro-level factors influencing the development of innovative drugs. Particularly for innovative therapies such as gene therapy and cell therapy, the regulatory framework often lags behind technological advancements. Taking the FDA's accelerated approval pathway as an example, while it lowers the threshold for drug market approval, it has also sparked ongoing debates regarding the strength of evidence required.
IV. Exploring the Path Across the "Valley of Death"
In response to the formidable challenges posed by the "Valley of Death," academia, industry, and regulatory bodies have conducted systematic explorations from various perspectives.
4.1 Process Reengineering: A Paradigm Shift from "Serial" to "Parallel"
BMS's reform practices in the 1990s remain a classic example of process innovation to this day. Addressing the primary cause of delays—the disruption in compound supply—BMS completely abandoned the post-development process scale-up approach and adopted a "proactive process research strategy": during the final six months of the lead compound optimization phase, process chemists intervened early to evaluate and determine the appropriate salt form and polymorph suitable for scale-up before the final candidate molecule was identified. This approach ensured that the active pharmaceutical ingredient (API) required for GLP toxicology studies was ready upon formal project initiation, eliminating the months-long gap period for process exploration.
BMS's experience demonstrates that deep cross-departmental integration is key to overcoming the "Valley of Death." The company established a "Development Coordination Team (DCT)," a multidisciplinary matrix organization led by frontline researchers, with members spanning ADME, drug safety evaluation, and clinical supply chain departments. The team is granted extensive tactical decision-making authority, with its sole mission being to develop comprehensive timelines and proactively allocate resources. This reform significantly shortened the FS-to-FIM (First Synthesis to First Human Administration) cycle from an average of 35 months, with the most successful projects completed in as little as 367 days.
4.2 Technical Empowerment: Extreme Optimization of the DMTA Cycle
Drug development is fundamentally an experimental science, centered around the Design-Make-Test-Analyze (DMTA) feedback loop. In 2009, AstraZeneca first broke down the lead compound optimization process into four DMTA steps and reduced the average delivery time from 23 days to 13 days through systematic optimization. After years of iterations, the average cycle duration of a DMTA loop was shortened by 46%, and the average cost of candidate compounds entering the safety evaluation phase was reduced by more than half.
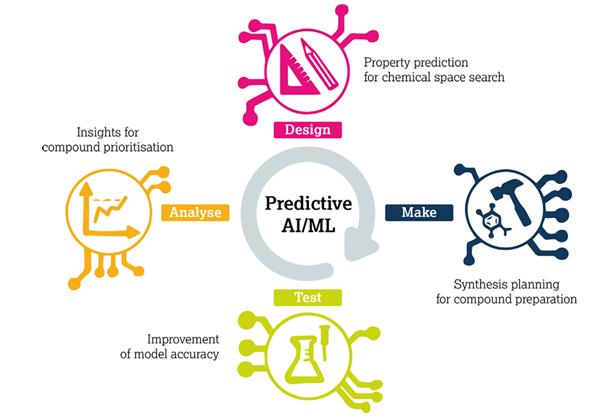
Figure 5 The Enhanced Design-Synthesis-Testing-Analysis (DMTA) cycle. The "Design" phase enhances performance by leveraging prediction scores for the activity and properties of individual components to drive chemical space exploration algorithms. The "Synthesis" phase improves efficiency through inverse and forward synthesis predictions that guide compound preparation. The "Testing" phase refines outcomes by prioritizing target compounds based on prediction results. The "Analysis" phase further enhances the process by employing predictive modeling to screen compounds for subsequent DMTA rounds.
In recent years, the emergence of new tools, algorithms, and technologies has empowered every stage of the DMTA cycle. Structural prediction tools such as cryo-electron microscopy and AlphaFold have enhanced the efficiency of obtaining target structure information; DNA-encoded compound libraries (DELs), combined with intelligent quality control, enable high-throughput screening of compounds; cloud computing has made computational evaluation of ultra-large-scale compound libraries feasible.
4.3 Capital Synergy: Patient Capital and Diversified Exit Strategies
The long-term nature of innovative drug development underscores the importance of "patient capital." As Lu Gang, investment partner at Chuangdongfang Capital, noted: "Biopharmaceutical investments have extended cycles, typically spanning over a decade from R&D to market launch. Investors must endure prolonged waiting periods and should not pursue short-term returns."
As China's innovative drug industry gradually enters a phase of tangible results, license-out is becoming a crucial pathway to overcome the "valley of death" in capital investment. By 2025, China's innovative drug exports achieved a historic breakthrough. As of December 31,2025, the total annual license-out amount for biopharmaceutical companies reached $136.555 billion, an increase of $84 billion or 160% compared to 2024; there were 157 transaction deals, up 63 from the previous year, representing a 67% growth. These transactions provided critical financial support and clinical development resources for early-stage research projects.
4.4 Scientific Origin: The Closed-Loop Construction of Translational Medicine
To fundamentally overcome the "Valley of Death," it is essential to enhance translational capabilities at the scientific foundation. Professor Cai Qingqing from Sun Yat-sen University Affiliated Tumor Hospital emphasized that improving the clinical relevance of target validation is key to shortening the translational cycle. Traditional animal models and cell lines often fail to fully replicate the tumor microenvironment in real patients; instead, greater reliance should be placed on large-scale multi-omics clinical samples and patient-derived organoid models, combined with technologies such as spatial omics and single-cell sequencing, to evaluate the role of targets across different patient subpopulations.
Establishing a bidirectional closed-loop system between "research and clinical practice" is equally crucial. Scientific findings must be promptly validated in clinical sample cohorts, while molecular data from patients in clinical trials should feed back into basic research. This closed-loop mechanism enables novel drug development to avoid unnecessary detours.
sum up
We must clearly recognize that the "Valley of Death" has never truly vanished; it merely continues to transform its form. While the challenges of drug development for small-molecule drugs have been overcome, novel therapies such as antibody-drug conjugates (ADCs), cell therapies, and gene editing present entirely new translational challenges. Meanwhile, as traditional issues of efficacy and toxicity are better managed, the multi-target networks of complex diseases, the heterogeneity of patient populations, and the interpretation of real-world evidence have emerged as new uncharted territories. As the widely circulated adage in the field states: "Biology is far more complex than we imagine."
The reason why the "Valley of Death" deserves to be repeatedly documented is not because it symbolizes despair, but because it defines the pinnacle of hope. Looking back in the spring of 2026, those formidable barriers that once led to the failure of 90% of candidate drugs are being gradually overcome by generations of pharmaceutical researchers through science, wisdom, and unwavering conviction. In the future, as artificial intelligence plays a more profound role in target discovery, organoid models more accurately simulate the human body environment, and regulatory frameworks become more flexible to accommodate innovative therapies, we have every reason to believe that the journey across the "Valley of Death" will ultimately evolve from a high-stakes survival challenge into a predictable and replicable scientific pathway.
The road ahead remains long, yet the direction has never been clearer. As illustrated by the bidirectional transformation framework—from laboratory to clinical practice, and back from clinical practice to laboratory—each closed-loop cycle narrows the gap between basic research and patient benefits. Those who dare to venture into the "Valley of Death" will ultimately receive the rewards befitting innovators on the other side: drugs that redefine the course of diseases, and countless lives rekindled.
For the ultimate purpose of traversing the 'Valley of Death' has never been mere arrival, but rather to free more people from confronting darkness alone.
References
1.Improving Translational Paradigms in Drug Discovery and Development
2.Why 90% of clinical drug development fails and how to improve it?
3.Bridging the Valley of Death The emerging imperative of Translational Science in the development of new drugs
4.Translational Challenges From Nonclinical to Clinical Program: Case Study Examples
5.How to deal with the unreliability of early data in new drug development
6.New Drug Development Accelerated! Decoding BMS's Classic Strategy: The Acceleration Logic from FS to FIM
7.Augmenting DMTA using predictive AI modelling at AstraZeneca
Disclaimer: This article is intended solely for knowledge exchange, sharing, and educational purposes only. It does not constitute commercial promotion and should not be regarded as medical guidance or medication recommendations. In case of any copyright infringement, please contact us for removal.
Our product recommendations:
1.1422140-08-6 https://www.bicbiotech.com/product_detail.php?id=6542
2.955-15-7 https://www.bicbiotech.com/product_detail.php?id=6543
3.1321111-54-9 https://www.bicbiotech.com/product_detail.php?id=6544
4.94876-25-2 https://www.bicbiotech.com/product_detail.php?id=6545
5.5450-40-8 https://www.bicbiotech.com/product_detail.php?id=6546




