With the rapid development of healthcare technology, the human life expectancy is gradually increased, and the aging population is also increased, which makes the incidence of neurodegenerative diseases (ND) increase. However, traditional animal models and 2D cell culture models have many deficiencies in simulating human pathophysiological conditions, such as failure to accurately simulate complex pathological lesions and neglect of multiple cases.
Given these limitations of the existing models, there is an urgent need to develop more advanced in vitro models. These models should be able to accurately replicate a variety of physiological conditions, multiple cell types and cell interactions, as well as the capability of real-time monitoring and regulation. Through such a model, we can study the pathological process of ND more deeply and provide more effective strategies for the prevention and treatment of the disease.
Recently, several alternative culture systems such as 3D cell culture, tissue engineering for 3D bioprinting, and microfluidic techniques have emerged as alternatives to conventional in vitro models to provide a more reliable and representative human system. Among them, microfluidic technologies can replicate key elements of organs and precisely control biochemical and biomechanical aspects, providing high-throughput, physiologically relevant and cost-effective solutions for disease research. By combining advanced biological and engineering technologies, chip-based in vitro modeling is expanding its possibilities, such as simulating ND simulation using 2D and 3D microfluidic chips.
Researchers from Sungkyunkwan University (Sungkyunkwan University) and other institutions recently discussed a general strategy for building ND models using microfluidic chips and introduced the next generation of advanced related models, mMS reported. Finally, the researchers provide an overview of the application of these models in academic and industrial drug development. Overall, the integration of microfluidic chips, stem cells, and biotechnological devices promises to provide valuable insights into biomedical research as well as the development of ND diagnostic and therapeutic options. Related papers were published in the Nature Communications journal under the title "Neuropathogenesis-on-chips for neurodegenerative diseases".
An ND model construction method based on a microfluidic chip
(1) Selection of the cell source
The human brain is composed of numerous neurons and non-neuronal cells, which forming a complex neuronal network. Cerebral vascular cells play a key role in the disease. Each brain cell has a different subtype, demonstrating its complexity. Limited access to the human brain requires the development of source cell methods with region-specific features, which is particularly important in ND modeling. Existing in vivo and in vitro animal models of ND provide valuable insights, but have limitations. External methods and non-mammalian species were also used in ND studies. Human embryonic stem cells (hESC) and induced neural differentiation methods are also used to simulate ND, but there are ethical issues and other aspects that require improvement. Familial and sporadic patient-derived induced pluripotent stem cells (iPSC) are another candidate cell source that produces pathological phenotypes associated with humans. Despite the important role of animal models, human patient-derived cells are more likely to be reliable in vitro models for ND studies.
(2) Select the microfluidic chip design
The chip was first applied in the brain study in the "Campenot chamber" system to study the effects of nerve growth factors on axon outgrowth. Subsequently, the researchers transformed the complex features of the brain into simple and miniaturized systems by simulating cells at different levels of the brain. The microfluidic chip consists of multiple compartments for cell co-culture that are connected by microchannels, porous membranes, and phase waveguides, allowing direct or indirect interactions between different cell populations. This chip design can control neurite growth direction, circuit connectivity, and size, and create different types of neuronal connections. Moreover, the on-chip valve and external factors can be used to control fluid flow and cellular environment, thus simulating chemical gradients of different concentrations, with an important role in disease modeling. The porous membrane-based chip serves as an interface between compartments to enables indirect and direct interactions to simulate structures such as the blood-brain barrier. These designs provide more dimensions of interaction that facilitate insight into the complex features of the brain.
(3) Monitoring and analysis steps
Microfluidic chips have several advantages in monitoring and analysis, such as real-time monitoring, multi-omics analysis, and integrated biosensors. Most microfluidic chips are made from transparent optical materials, introducing very small amounts of staining dyes and antibodies through the fluid channel entrance, but preventing contamination and bubbles. Integrated biosensors can continuously measure cellular properties and assess cellular responses. The chip brain measures blood-brain barrier integrity and neuronal electrical signals. Out-of-chip analysis tools can be used to analyze samples. Open chip design offers more advantages, such as exposure to chemical composition, simplified collection and analysis, but should pay attention to the controllability of fluid flow.
(4) Application of modeling of ND pathogenesis
To study neurological related diseases, relevant anatomical and physiological units can be replicated by matched chip design. However, existing microfluidic chip studies are limited by cell origin and 2D static design and lack physiological relevance. The 2D system fails to mimic complex interactions and physiological conditions that affect cell function and in vivo relevance. In contrast, 3D extracellular matrix (ECM) gels provide a more realistic cellular environment, with improved cell morphology, migration, and signaling, allowing for time-dependent differentiation and neurodegenerative changes. To create the 3D microenvironment, properties of the ECM that influence brain cell behavior and disease mechanisms. Hydrogels have been integrated into microfluidic chips to mimic a more physiologically relevant brain tissue environment.
And ND chip application cases
(1) Alzheimer's disease (AD) chip
Microfluidic chips have been widely used to study the pathogenesis of AD, including A β and tau lesions, mitochondrial dysfunction, and neuroinflammation, providing new insights that could not be obtained by traditional culture methods. The chip can visualize protein lesions in real time and discover the propagation properties of tau. Meanwhile, microfluidic chips can mimic in vivo dynamic conditions such as interstitial flow velocity and A β aggregate gradient formation to study the toxic effects of A β on neurons. In addition, the chip was used to study human neural progenitors transduced with familial AD specific mutations, as well as the role of astrocytes and pathogenic proteins in neuroinflammation. By integrating different cell types, the chip can reveal the molecular mechanisms of neuroinflammation. In recent years, studies have also focused on the role of brain features such as the BBB and neurovascular units in the pathogenesis of AD. The combination of microfluidic chips with 3D culture, as well as lymphatic flow simulations, offers more possibilities to explore similar conditions in vivo. These studies are expected to provide new clues on drug screening and treatment strategies for AD.
(2) Parkinson's disease (PD) chip
Most of the existing PD chip studies have focused on α -syn-related pathogenesis, but few studies involving dopaminergic neurons. The investigators used a microfluidic chip to simulate dopaminergic neuronal apoptosis and α -syn propagation in PD. The chip design enables a clear observation of α -syn fiber uptake and its transport between neurons. The toxicity of the α -syn oligomers to the neurons of the mutant PD patients was also explored. In addition, the uptake and diffusion of α -syn were successfully demonstrated by a chip controlling fluid dynamics with microvalves. The multi-compartment chip can simulate the propagation of α -syn between neurons and successfully reproduce the complex neural networks. Microfluidic chips are also applied to study the impairment of dopaminergic neurons in PD, the role of the BBB in PD pathogenesis, and the relationship of astrocytic activity with BBB dysfunction and inflammation.
(3) Amyotrophic lateral sclerosis (ALS) chip
Based on newly discovered genetic factors, the investigators developed rodent models to study ALS that mainly reflect familial ALS and reveal dysfunction of cellular processes associated with ALS in vitro. Although microfluidic chip models for studying the aggregation and dissemination of toxic proteins already exist, no specific model for sporadic ALS. Neuroinflammation is a pathogenic feature of ALS, and microfluidic culture systems can be used to model the effects of astrocytes on neurons. Using the compartmentalization elements of the microfluidic chip, the investigators co-cultured myocytes and motor neurons to study NMJ damage in ALS. These models reproduce certain features of ALS but may lose physiological relevance when using human-specific motor neurons and primitive rodent myofibers. One of these custom 3D microfluidic chips was able to co-culture patient-derived motor neuron spheroids and skeletal myoblasts and successfully recapitulate some of the pathological processes in ALS.
(4) Huntington's Dance Disease (HD) chip
Microfluidic chips have had relatively few applications in HD research. These chips mainly simulate the corticostriatal network, which is vulnerable in HD. Studies using transgenic mouse neurons to explore the toxicity of mHTT on neural circuits. The chip design allows the observation of striatal neuronal degeneration and reduced synapses, as well as transport defects due to mHTT. The team also used chips to study drug targets, such as the APT 1 inhibitor ML348, which ameliorates motor deficits and behavioral changes. In addition, the five-chamber chip mimics the basal ganglia circuit and showing connections between different cell populations. The microfluidic chip integrating MEA can analyze the neural network function in real time and detect the effect of Huntington protein on the circuit. Electrical stimulation microfluidic chip is one of the directions of improvement that reproduces and measures neuronal activity in corticostriatal circuits, but has not been applied to HD studies.
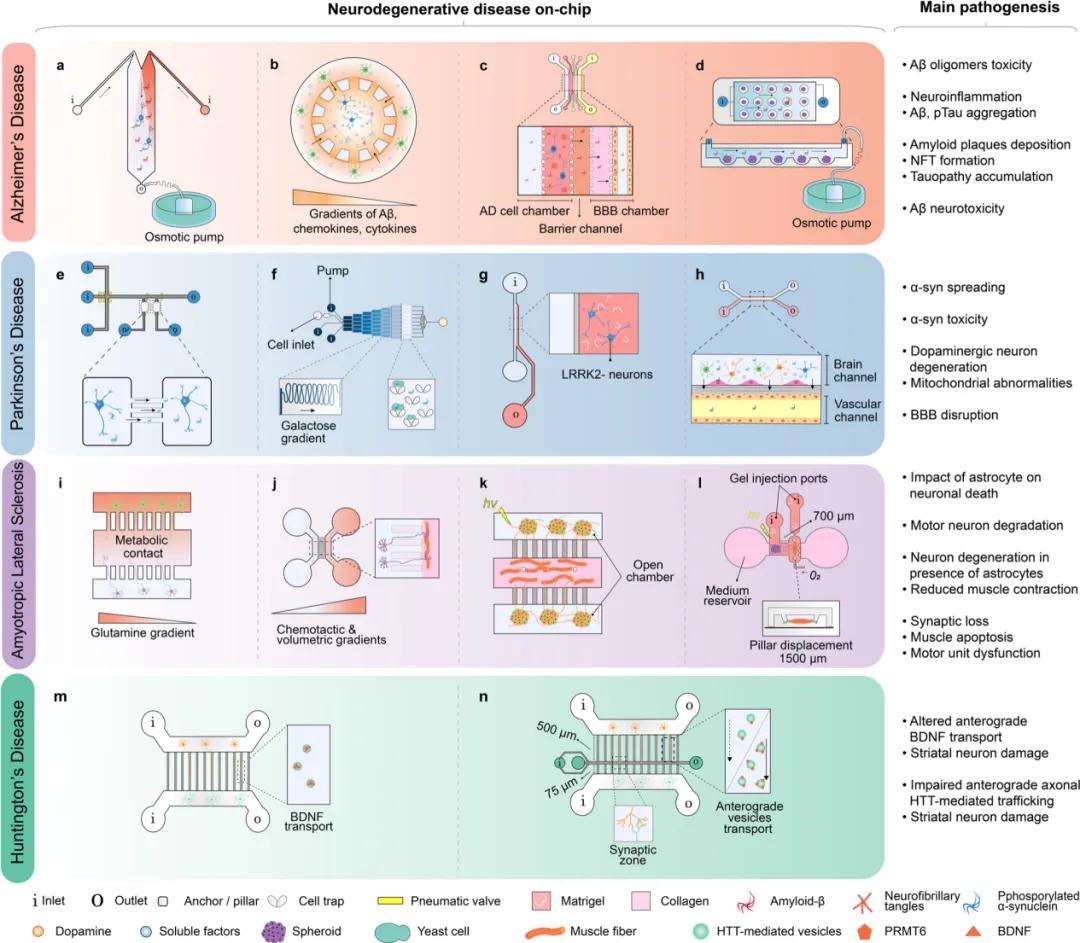
Example of a microfluidic chip used for ND modeling
In vitro 3D model based on a microfluidic chip
Brain organoids can mimic disease-specific brain regions with different cellular compositions, such as the forebrain, midbrain, and hindbrain. The investigators use multiple methods to simulate ND, but are mostly limited to familial cases. For the interactions between different brain regions in ND, Pasca et al. created combined organoids (brain assemblers) to better generalize the regional interactions and apply them to a variety of disease models. The fusion cortical vascular aggregate, that mimics the accelerated effect of systemic inflammation on AD neuroinflammation, was developed by Kang et al. Rickner et al. generated neural-astrocyte assemblies that showed some pathological changes and responded to treatment. However, organoid models remain controversial in studying ND and are currently more suited to mimic neurodevelopmental disorders, but their well-established results in cellular composition, anatomical features and brain convolution are also encouraging. Recently, multi-variety organ culture platforms have been designed to improve the in vivo characteristics, maturity and uniformity of organoids while reducing the cultivation time and amount of labor. These platforms have the ability to cooperate with immune and vascular components and effectively effectively with bioimaging and biosensors. Some studies have solved the problems in the traditional organoid culture schemes through the "one-stop" microfluidic platform, and realized the full automation of cultivation, maintenance and monitoring. Moreover, scaffold materials and microfluidic chip design in 3D culture also play an important role in promoting the maturation and characteristic development of organoids. Meanwhile, simulation of vascularization and immune interactions is crucial for further characterization of brain organoids. Microfluidic chips can also be integrated with bioimaging devices and biosensors for accurate monitoring and analysis.
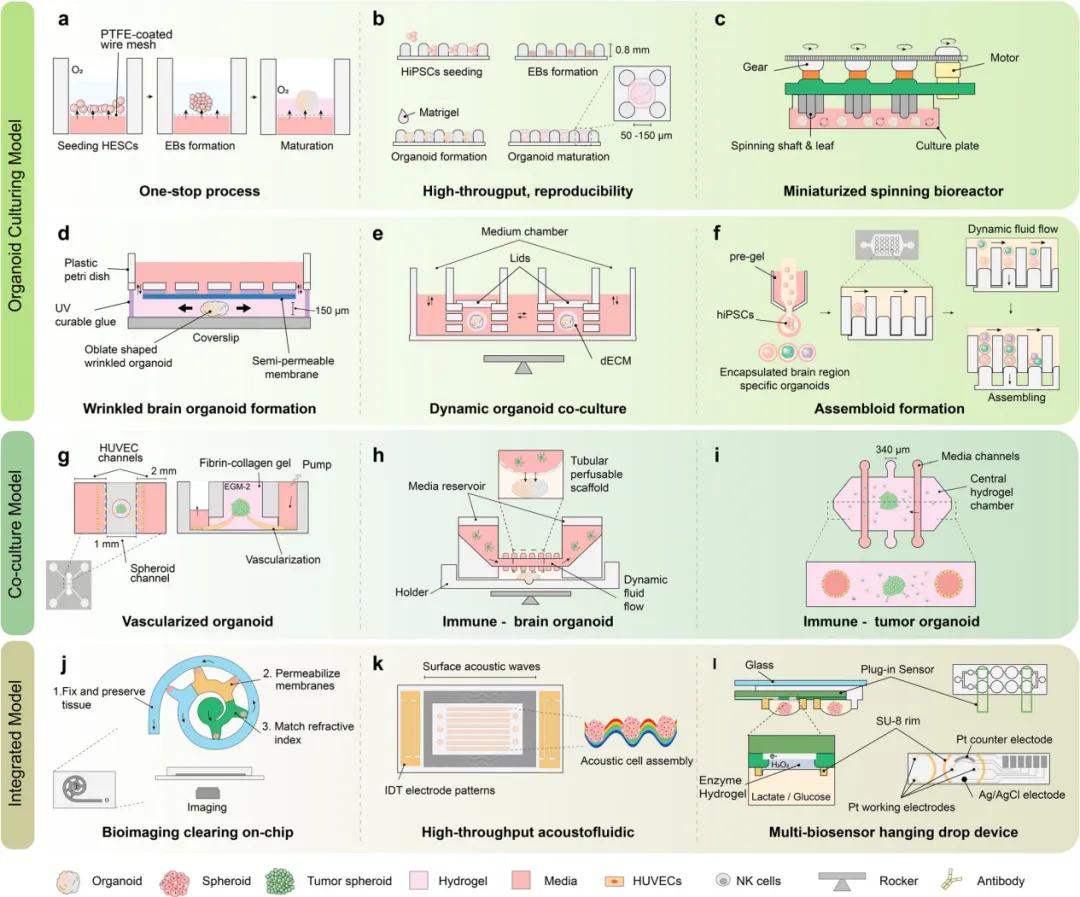
Representative supraasine organoid design
Application prospect of microfluidic chip-based in vitro models in the field of pharmacology
(1) Challenges faced by ND drug development
Despite substantial progress in disease understanding and technological innovation, the development of effective treatments for NCDs remains challenging. There have been recent breakthroughs in AD treatment, but the effectiveness of new drugs is only for specific stages, and the effect of A β -antiantibodies in human cognition is controversial. Considering the multifactorial and heterogeneity of ND, more efforts are still needed to expand drug effectiveness. Furthermore, the development process is cost-effective and time-efficient. In the past, AD drug development has invested heavily with limited results. Reasons for failure include improper timing of intervention, insufficient biomarkers and inaccurate research models. Familial genetically engineered mouse models have limitations that cannot fully replicate ND features and long-term preservation is expensive and time consuming. Thus, shifting research to more physiologically relevant in vitro models of human cells combined with new drug characteristics is expected to reduce cost and time and produce more representative test results.
(2) Application of microfluidic chips in the pharmaceutical industry
In the last two decades, in vitro models have gradually developed in academia, but microfluidic chips have not been adopted in the pharmaceutical industry. As FDA policy shifted to drug testing using more human-related models, academia and the pharmaceutical industry began to use organ-on-chip and organ-based models. Some large pharmaceutical companies have used organoids and microfluidic chips to improve drug screening accuracy. Global efforts are also emerging to translate academic results into the pharmaceutical industry, utilizing standardized and reproducible drug screening methods. The NIH launched a grant program to validate tissue microarray platforms for drug testing, including microphysiological systems reproducing the kidney, liver, and blood – brain barrier, to improve the reliability, reproducibility, robustness, and throughput of their applications.
(3) Microfluidic chip used for academic-level drug screening
The pharmaceutical industry makes continuous efforts to address the issues of reproducibility and scalability, while the academic field also conducts drug screening and toxicity studies on potential ND drug components. Many companies offer microfluidic chips for researchers to use in disease mechanisms, drug screening, and toxicity studies. Several groups have used these chips to re-evaluate previously rejected drug candidates. Furthermore, monolayer culture methods are often used for drug discovery, but 3D cell culture and patient-derived organoids are thought to provide models closer to in vivo features and are expected to improve clinical predictivity. Academics have begun to apply organoids to drug discovery studies that assess not only the effects of drugs on functional 3D co-cultures, but also the delivery of drugs through the endothelial cell (EC) barrier. The novel organoid culture drug screening platform also demonstrated the potential of patient-derived organoids in ND drug screening.
(4) The application prospect of advanced microfluidic chips in the pharmaceutical industry
Global collaborative efforts to integrate microfluidic chips and advanced technologies are expected to revolutionize the pharmaceutical industry. Organoid chips and multi-organ on-chip systems play a key role in drug discovery, from target identification to preclinical screening and can even be used for effective therapy in clinical stage-determining patient subtypes. However, achieving this goal requires overcoming technical and industrial challenges such as reliability, reproducibility, and compatibility, while defining validation and modeling benchmarks. Advanced chip systems can validate the effects of drugs on neuropathology, measure functional changes, and provide a clinically relevant assessment of ND. Multi-organ chip platform can test the mechanism of action and safety of drugs, reduce the dependence of animal models, and provide more representative, cost-effective and accurate results. Patient-derived cell sources may facilitate effective therapeutic development. In the future, microfluidic chips will contribute to brain organoid and assemblies translation and ND drug discovery and personalized medicine development, and despite biological, technological and commercial challenges, microfluidic chips and molecular biophysical technologies are expected to promote the understanding of ND pathogenesis and reduce reliance on animal models.
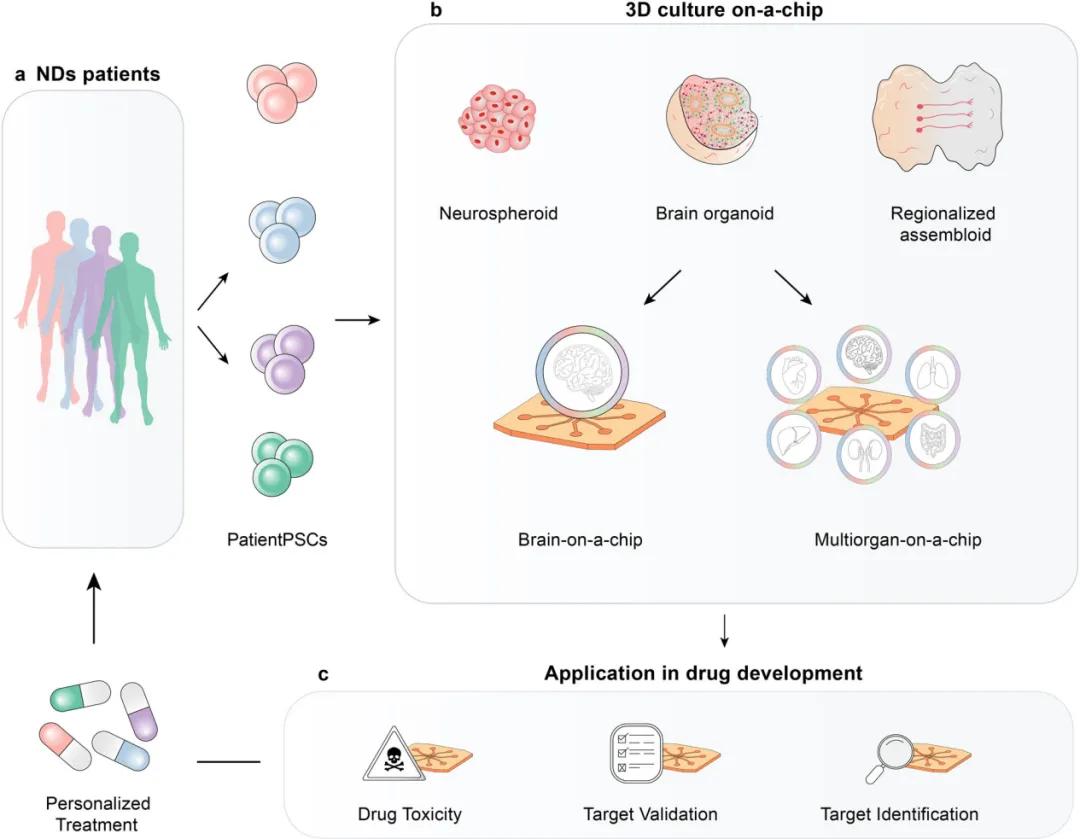
Future perspectives for on-tablet ND for drug development
In conclusion, this paper explores the use of advanced in vitro systems to simulate different types of neurodegenerative diseases, providing valuable insights for biomedical research and drug development. Traditional microfluidic chips under 2D and static conditions have physiological limitations when simulating neurodegenerative diseases, while adopting 3D extracellular matrix gel can provide more mechanical structure and biochemical signals of cells, thus improving the correlation in cell morphology, migration behavior, signaling and gene expression. Furthermore, the pathophysiological mechanisms of neurodegenerative diseases, such as the dissemination and aggregation of Tau, and the importance of the extracellular matrix for the development and progression of the disease are mentioned. This review helps readers to gain a better understanding of recent developments and future directions in the field of neurodegenerative diseases.
Paper link:
https://doi.org/10.1038/s41467-024-46554-8




