For the drugs in development, the clinical focus is on the safety and effectiveness of the research product, while the pharmacy focuses on the stability and uniformity of the research product. Speaking of dosage forms, solid preparations pay more attention to the crushing and mixing of raw materials and accessories, while liquid preparations pay more attention to the dispersion and dissolution of raw materials. Whether it is the crushing and mixing of solid, or the dispersion and dissolution of liquid, it directly affects the stability and uniformity of products, so the stability study of products is very important and is an eternal topic in the study of pharmaceutical preparations. Therefore, this paper focuses on the introduction of the drug stability study.
The stability test of drug preparation is the change of product quality under the influence of temperature, humidity, light, vibration, use and time, mainly including the stability of influencing factors, acceleration stability, long-term stability, transportation stability and use stability. The investigation results of product stability can provide scientific basis for the production, packaging, storage, transportation and use of drugs, and can also provide reference and support for the formulation of product validity period in the later period.
1. Influence factors
In the drug development stage, the investigation of influencing factors is particularly important. Because the influencing factors are investigated under relatively intense conditions, the stability difference between different prescription processes can be distinguished in a short time, so as to save the R & D time and improve the R & D efficiency. At the same time, it can provide reference for long-term and accelerated selection of temperature and humidity stability, and also provide support for product impurity research and method analysis in the later stage.
High temperature, high humidity and light are the common investigation items of influencing factors. Among them, high temperature is the most commonly used investigation item, according to the characteristics of the preparation, 40℃ and 60℃ can be selected. Generally, 40℃ for refrigerated drugs; 60℃ at room temperature: 5 days, 10 days, 30 days, 7 days, 14 days and 30 days according to personal conditions. Hygroscopic powder or tablets need to investigate the impact of humidity on the product, can choose 25℃ / RH 90% or 25℃ / RH 75%, two conditions can be investigated at the same time, the general inspection time is 5 days and 10 days. It should be noted that because the liquid preparation is a sealing system, the influence of humidity may not be investigated.
Regardless of the dosage form, light investigation is inevitable. Light investigation can be divided into fluorescent lamp, strong light. The light intensity of fluorescent lamp is 200-300lux, which mainly provides basis and support for the development of mass production and clinical administration. The strong light light intensity is 4500 ± 500lux, which can provide a reference for the study of product light stability and impurities. The inspection time can be 5 days, 10 days, 30 days, or 7 days, 14 days, 30 days according to personal circumstances.
Low-temperature cycle, freeze-thaw cycle, placement conditions, shaking and oxidation can be selectively investigated according to the characteristics of the preparation. Considering the possibility of overtemperature in drug use or storage, the low temperature and freeze-thaw cycle should be investigated for refrigerated drugs. Considering the influence of packaging material, liquid or lyophilized preparations generally need to investigate the placement conditions, such as upright and inverted. Considering the influence of vibration, the vibration stability is needed to investigate the unstable drugs, for example, protein drugs. See Figure 1 for the common investigation items of influencing factors.
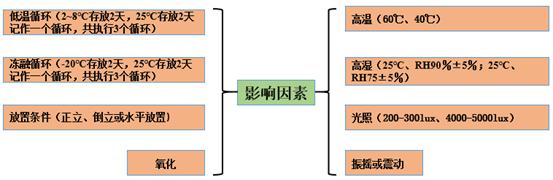
Figure 1 Common investigation conditions of influencing factors
2. Accelerated stability
Accelerate stability mainly examines the influence of temperature and humidity on drugs, its conditions are lower impact factors, higher than normal storage requirements, the purpose is to accelerate the chemical or physical and chemical changes of drugs, study the stability of drug preparation, to provide necessary data and data support product prescription design, process improvement, quality research, packaging design, product transportation and storage. Before conducting accelerated stability studies, temperature and humidity conditions should be selected according to the properties and characteristics of different preparations. Accelerated test conditions of common preparations are shown in Table 1.
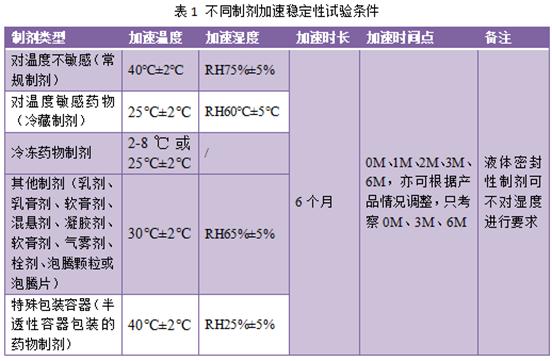
Generally, for drug preparations that are not sensitive to temperature, the experimental conditions: temperature 40℃± 2℃, relative humidity 75% ± 5% conditions were investigated for 6 months. Detection points can be 0M, 1M, 2M, 3M and 6M, or can be adjusted according to the product situation, only 0M, 3M and 6M.
The temperature sensitive drug preparations are expected to be refrigerated (2-8℃), the accelerated test conditions for such drugs are 25℃± 2℃, relative humidity 60℃± 5℃ (the humidity test is not required for liquid preparation), the test time is also 6M, the test time point is the same as the above.
For emulsions, cream, ointment, suspension, gel, ointment, aerosol, suppository, effervescent granules or effervescent tablets, the acceleration conditions should be conducted under the test conditions of 30℃± 2℃ and relative humidity of 65% ± 5%, and the investigation time and time point are the same as the above.
For pharmaceutical preparations packaged in semi-permeable containers, such as plastic ampoules, low-density polyethylene bottles, and ophthalmic preparation containers, accelerated conditions should be tested at a temperature of 40℃± 2℃ and a relative humidity of 25% ± 5%. The inspection time and time point are the same as those mentioned above.
3. Long-term stability
Long-term tests are carried out under the actual storage conditions or close to the actual storage conditions of the drug, whose purpose is to provide a basis for the formulation of the validity period of the drug. As with accelerated stability studies, temperature and humidity conditions should be selected according to the properties and characteristics of different preparations before conducting long-term stability studies. Long-term test conditions of commonly used preparations are shown in Table 2.
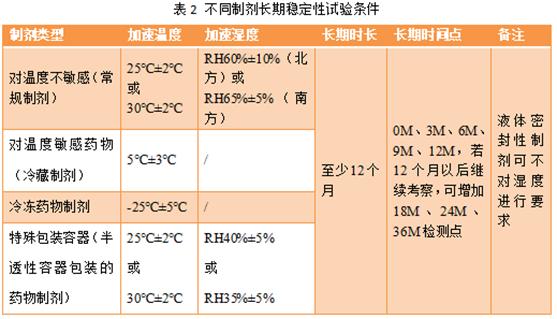
China is vast, with the difference in temperature and humidity between north and south. There are common conditions for long-term test: temperature 25℃± 2℃, RH 60% ± 10%, 30℃± 2℃ temperature and RH 65% ± 5%. The placement time should be at least 12 months, and the sample should be taken once every 3 months, so the detection points are 0M, 3M, 6M, 9M and 12M. If the investigation continues after 12 months, the detection points of 18M, 24M and 36M can be added.
For temperature-sensitive drugs, attention should be paid to the choice of their long-term test conditions. Drugs intended for the refrigerated storage can be tested at a temperature of 5℃± 3℃; drugs intended for the frozen storage can be tested at-25℃± 5℃.
For pharmaceutical preparations packaged in semi-permeable containers, long-term conditions shall be tested at 25℃± 2℃, 40% ± 5% or 30℃± 2℃ and 35% ± 5%. The inspection time and time point are the same as those mentioned above.
4. Transportation stability
Any drug preparation will face transportation problems after leaving the factory, so the transportation stability study should be carried out. In the preliminary study, the shaking conditions can be used to simulate the transportation of the product stability, so as to know whether the handling, shaking and shaking are stable during the transportation in advance, which can provide reference for the subsequent study of product transportation stability. With the approaching of the marketing time, it is necessary to carry out the real transportation distance, vibration amplitude and frequency according to the market situation of the product. If refrigerated drugs, the impact of partial temperature problems such as cold chain removal on drug preparations should be investigated.

5. Use of stability
Studies of influencing factors, long-term and accelerated tests were conducted in unopened or unused drugs, which cannot understand the product stability during drug use, so the use stability study is needed. For sterile lyophilized powder, the reconstitution time, compatibility stability and use duration should be investigated. If the administration time exceeds 4h, the product microorganisms should be evaluated. For multi-dose products such as insulin, the use stability study should be carried out according to the clinical use time and puncture times, and we should ensure that all physical and chemical, related substances, microorganisms and other indicators meet the requirements during the use period.

6. Summary
Stability research runs through the whole life cycle of the product, providing effective data support and scientific basis for the development, production, packaging, storage, transportation, use and validity period formulation of drugs, and has a crucial impact on the safety and effectiveness of drugs. Therefore, when carrying out the stability research, it is necessary to carefully consider the proposed investigation conditions to ensure that the selected investigation conditions are representative. Only when the conditions are representative, the data will be representative, which can provide a scientific basis for the safety, effectiveness and stability of our products.
reference documentation:
[1] Technical guidelines for chemical drug stability research.
[2] Technical guidelines for the stability study of biological products (trial).
[3] Pharmacy People's Health Press.
[4] Licensed Pharmacist examination Pharmacy professional knowledge (I) China Medical Science and Technology Press




