Of the various routes of drug delivery to the human body, the oral route is most preferred due to patient compliance. Drug delivery is possible using different dosage forms, such as tablets, liquids (solution, suspension, etc.), and capsules. Solid dosage forms account for about two-thirds of all available dosage forms. In the development of new drugs, the choice of dosage forms is mostly ordinary oral solid preparations. Pelleting is one of the most important unit operations in the production of pharmaceutical dosage forms. In the pharmaceutical industry, granulation is the mixing of powder mixture (usually composed of drugs and excipients) and by dry pressure method or through the use of adhesive solution, called dry and wet particles, respectively. In addition to the common granulation process, with the increasing variety of excipients, powder direct pressure / powder direct irrigation process is also active; of course, with the emergence of the physical and chemical properties of poor drugs, innovative technologies, such as spray drying or hot melt extrusion to prepare amorphous solid dispersion. According to the physical and chemical properties of the API, how to choose the appropriate production process, the production classification system MCS may be used to answer this question. According to the production classification system (MCS), the MCSI-production process steps (as shown in figure 1), complexity and cost is gradually increased, but bear the API bad physical properties (mechanical properties) ability gradually increase, at the same time bear API bad chemical properties (damp and heat stability) ability reduced, for the risk of API physical and chemical stability.
The Production Classification System (MCS):
(Ⅰ)Direct Compression(DC)
(Ⅱ)Dry Granulation(DG)
(Ⅲ)Wet Granulation(WG)
(Ⅳ)Other Technologies(OT)
Powder direct pressure / direct irrigation: powder direct tablet pressing or powder direct capsule, contains only two main steps, refers to the drug substance and appropriate excipients (filler, disintegrant, lubricant, etc.) after evenly mixed with tablet compression or capsule. Compared with dry grain and wet grain, the process is simple. This type of process the strongest dependence on the properties of API, API properties is not only need to be acceptable, but these properties can maintain consistency, especially the powder of API, such as API particle size, shape and density properties change between each batch, will affect the uniformity of mixed particles, which may cause the product content and content uniformity problem. If necessary, standards for the particle size and size distribution of API are required to ensure the consistency of production products. Of course, other properties of the API, such as morphology, affect the quality of the product (fluidity of the intermediates, mixing uniformity). The formulation of the above standards depends on the actual nature of API in each project and its impact on product COAs, and then establish the limitation of API and excipients CMAs. In some cases, the particle size, morphology, density and other properties of the auxiliary materials also need to be further limited, specific situation specific analysis.
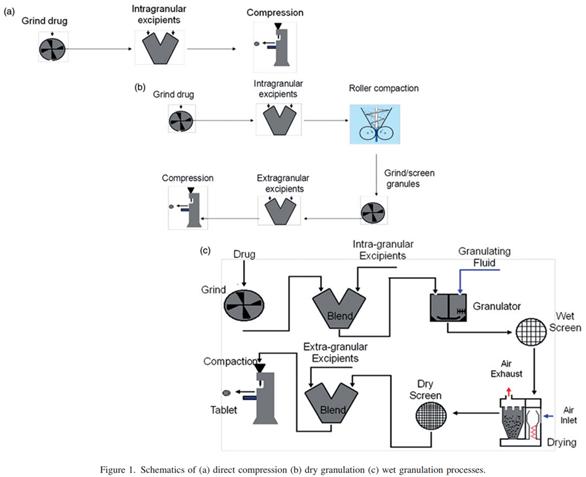
Figure 1 Process routes for three major general oral solid preparations (reference 1)
Absolute advantage analysis of powder direct pressure:
save resource
The powder direct sheet pressing method can not only ensure the stability of product quality, but also reduce the production cost and improve the economic benefit of enterprises.
Disintegration of the booster tablets
When the tablet is prepared by powder direct tablet pressing method, the disintegrant will not reduce the disintegration performance due to the early contact with water, thus ensuring good disintegration characteristics. In addition, due to the lack of pelleting, the tablet disintegration will not form large mass particles, but the formation of relatively large surface area of fine powder, can be better distributed in the body, conducive to the release and absorption of drugs.
solubilization
Using powder direct pressing process, choose good hydrophilic excipients (such as lactose) as a filler, on the premise of ensuring the tablet quickly disintegration, drugs together with lactose powder dispersed in water, then lactose dissolution and on the surface of the drug with van der waals force and water hydrogen bond, reduce the hydrophobicity of drugs, help to improve the dissolution of drugs.
Improve stability
Chemically unstable drugs are prone to hydrolysis in the process of wet particles, or are damaged by heat in the drying process. The preparation process of powder direct tablet pressing method does not require granulation and drying, and the drug is not affected by dampness and heat, which better protects the stability of the drug. Although powder direct pressure / direct irrigation has so many advantages, why is it still not used by more enterprises?
first, The excipients selected for direct powder pressure require good powological properties, Requires special and complex process preparation, Generally, the more expensive, Some companies and their leaders may be controlled by input, Powder direct pressure / direct irrigation as the final choice of the process; Once direct tablet / direct capsule is chosen, If a change of the process occurs in the subsequent development, Major changes are required, Clinical bridging studies may be required, Not confident enough in the process; The roots may lie in technical problems, For the powder chemistry control of the raw materials themselves (such as compressibility, fluidity, dilution potential, content uniformity, lubrication sensitivity, etc.), Production efficiency (such as compression speed) and production line utilization, etc.
Dry legal grain: first the drug powder and auxiliary materials, and then add the mixture to spiral feeding mechanism, powder through the spiral feeding mechanism to the roller area, in the roller area is pressed into pieces, discharge body in the whole by rotating blade broken become standard drug particles, complete the dry legal grain production process.
Feeding mechanism, roller mechanism, crushing mechanism and whole grain mechanism are the main working components of dry granulator, among which the feeding mechanism and roller mechanism are the core components of dry granulator. The drug powder is transported to the roller body through the feeding mechanism, and is pressed into the tablet body in the roller mechanism. The density and hardness of the discharging body determine the quality of the tablet body, and then determine the yield of the drug particle. The study of the movement process of powder in the feeding mechanism and the compression molding process in the roller mechanism is helpful to understand the influence of operating parameters, geometric parameters and friction coefficient on the roller molding of drug powder, so as to guide the design, production and production of dry granulator and the selection of operating parameters, and improve the production efficiency of dry granulation.
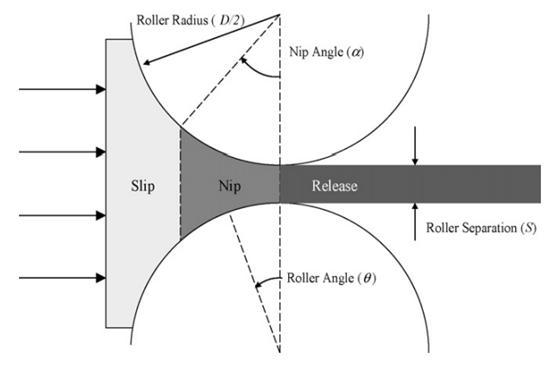
Figure 2 Diagram of dry powder (reference 2)
Johanson The movement of powder in the roller mechanism is divided into three stages, namely Slip (slip), Nip (engagement) and Release (release). The pressing process of powder is mainly realized in Slip and Nip. The drug powder is transported to the roller mechanism by the spiral feeding mechanism. When the movement speed of the powder enters the roller mechanism is less than the line speed of the roller surface (the powder is located in the Slip area), and the powder is driven forward by the roller. After a distance forward, the movement speed of the powder on the surface of the roller is consistent with the line speed of the powder enters the powder in the Nip area, and the discharging sheet enters the Release area to complete the pressing process. Refining the molding process of dry grain rolling, and find the key process parameters CPPs that affect the key physical and chemical properties CQAs of the ultimate particle.
Dry granules are often used in the pelleting of heat-sensitive materials, drugs that are easy to decompose in water and drugs that are easy to compress. The advantage of wet and dry legal particles is that they do not involve water and heat in the process, and are suitable for granulation of sensitive damp-heat materials. At the same time, the dry legal grain occupies a small space, suitable for continuous production. Most of the ideal properties for direct powder pressure also apply to dry rule particles, except for properties related to fluidity or heap density. Compared with powder direct pressure / direct irrigation, dry particles provide additional pressing step, and API is partially compacted with other excipients to form particles, with greater tolerance for the properties of API.
Of course, there are some small problems with dry particles: dry particles are not suitable for pressure or secondary pressure of poor materials; due to high pressure particles, high particle density, may reduce the dissolution of products; for pressure unstable API (such as pressure crystal), dry particles process can not be used.
Wet particles: During wet particles, particles are produced by adding a liquid / dry adhesive to the powder mixture. The drug is mixed with the excipients and processed using a solvent (aqueous or organic solvent) and subsequently dried and ground to produce the particles. The solvent mixed into the powder can form a sufficiently strong binding between the powder particles to lock them together. However, after liquid drying, some powder may disperse. In this case, a liquid solution containing the adhesive is required. When it comes to water-sensitive drugs or requiring rapidly drying particles, organic solvents are used, for example, ethanol. Furthermore, stabilizers, such as pH modulators in tight contact with drugs, can be used, which can maximize product stability. After removing or drying the solvent, the powder mixture forms more dense clumps. In traditional wet grain methods, soft materials are forced through a sieve to produce a predetermined size of wet particles, which are then dried. Subsequent post-drying, screening / grinding steps break the appendage into particles with the desired particle size and distribution.
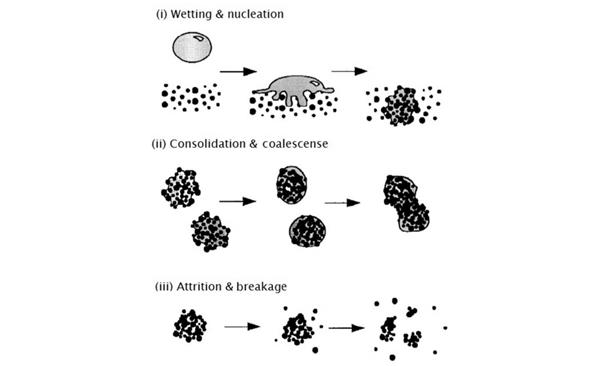
Figure 3 Wet granulation mechanism (reference 3)
The mechanism of wet-rule particle formation involves the following three processes:
a. Wetting and nucleation (Wetting and Nucleation)
The liquid adhesive is in contact with and distributed through the dry powder bed to produce a granular core. This mechanism is considered to be an important stage in the granulation process, however, it is rarely identified and separated from other effects (eexample, coalescence and abrasion).
There are two important processes in the nucleation zone. The first stage is nucleation, which is a function of the wetting thermodynamics and kinetics. The second stage is the dispersion of the adhesive in the powder mixture to ensure efficient mixing of the powder and the adhesive, which is a function of the process variable. Selection of undesirable combinations of powders and adhesives (e. g., high contact angles) or use of inefficient adhesive dispersion methods (e. g., high fluid flow rates or poor spray properties) produces products that are difficult to control and replicate.
b. Consolidate and grow (Consolidation and Growth)
At this stage, collisions between two particles, particles and excipients powder, or between a particle and the device lead in compaction and growth of the particles. Particle growth occurs whenever materials in the granuler collide and stick together. For two large particles, this process is traditionally called coalescence (coalescence), and the adhesion of fine materials to large pre-existing particles is often referred to as layered (layering).
These growth processes can be initiated upon the addition of the liquid to the stirred powder (i. e., simultaneously with the wetting and nucleation phases) and can continue after the liquid addition is complete. Whether the collision between two particles leads to permanent coalescence depends on multiple factors, including the mechanical properties of the particles and the availability of liquid adhesives on or near the particle surface.
c. Damage and wear (Attrition and Breakage)
At this stage, wet or dry particles are broken due to impact, shear, or compaction in the granator or during subsequent product processing. Breaking of wet particles can affect the final particle size distribution, especially in high-shear granators. In some cases, breakage can limit the maximum particle size or help to disperse the adhesive. On the other hand, the wear of dry particles leads to the production of dust-like fine particles.
To understand the mechanism of wet particles, we can better explain the difference between the formation mechanism in the wetting agent and the atomization in the size of the droplet is controlled by each droplet to form a nuclei; when the solution is added by dumping, the loose wet material mass, under the action of shear force, is scattered into small particles to form nuclei. This is only an explanation of the wet method of granulation machinery, and more content and applications need to be deeply explored, which is not the focus of this paper.
There are three main ways of wet grain: high speed shear granulation, low speed shear granulation and fluidigranulation, most commonly used for high speed shear granulation. High-speed shear mixing pelleting technology is made by the mixing paddle to make the material regularly three-dimensional movement, at the same time through the high-speed rotating cutting knife cut many times, so as to form a fine, uniform and round particles. High shear wet legal grain is the most commonly used method in the granulation process. Compared with other granulation technologies, the process is easy to enlarge and the particles are easy to mix. To facilitate particle size growth, the drug load of high shear wet legal particles is 50%.
However, compared with powder direct pressure / direct filling, dry particles, wet particles involves more steps, and the introduction of wetting agent (water or ethanol, acetone and other organic solvents), this process generally requires the addition of a large amount of water, such as 20~50%. After the wet legal grain, the prepared soft material needs to be dried, which brings heat, and the introduction of humid heat brings a lot of physical and chemical instability. Although water can be removed during drying, this production process still poses huge risks, especially for water-sensitive drugs.
Table 1. Reasons for Process Selection (Ref. 4)
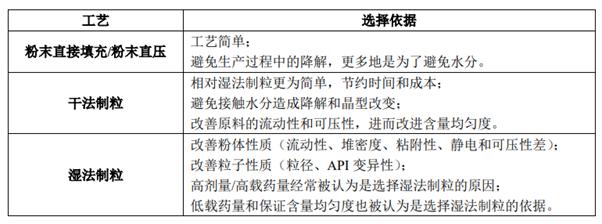
brief summary:
The development of solid dosage form is still the mainstream drug carrier in the pharmaceutical industry. The final realization of solid dosage form is inseparable from the robust preparation process, including powder direct pressure / direct irrigation, dry particles, wet particles and others. If the preparation development is compared to a big war, how to make the appropriate combat strategy and tactics to decide who can win. The ancient wisdom of our ancestors tells us: know yourself and know the enemy, and win a hundred battles. Like we need to develop a solid preparation, clearly know the nature of our API and master the technology, and the nature of the API and optional process pairing, this is a "friend", know you know, understand their task objectives, scientific analysis, finally perfect in the implementation, gradually grow in the perfect. Above small article, in to tell everyone! If there is something wrong, please tell me!
reference documentation:
1. Production process route selection of oral solid preparation based on drug substance properties
2. Experimental and numerical study of dry rule grain technology
3. Handbook of Pharmaceutical Wet Granulation Theory and Practice in a Quality by Design Paradigm (2018)
4. Production route of oral solid preparation based on drug substance properties: analysis of EU review report
5. Practical application of roller compaction process modeling




